NTA JEE Mains 8th April 2024 Shift 2
For the following questions answer them individually
NTA JEE Mains 8th April 2024 Shift 2 - Question 31
Identify the correct statements about p-block elements and their compounds. (A) Non metals have higher electronegativity than metals. (B) Non metals have lower ionisation enthalpy than metals. (C) Compounds formed between highly reactive nonmetals and highly reactive metals are generally ionic. (D) The non-metal oxides are generally basic in nature. (E) The metal oxides are generally acidic or neutral in nature. Choose the correct answer from the options given below :
NTA JEE Mains 8th April 2024 Shift 2 - Question 33
When $$\psi_A$$ and $$\psi_B$$ are the wave functions of atomic orbitals, then $$\sigma^*$$ is represented by :
NTA JEE Mains 8th April 2024 Shift 2 - Question 34
The equilibrium $$Cr_2O_7^{2-} \rightleftharpoons 2CrO_4^{2-}$$ is shifted to the right in :
NTA JEE Mains 8th April 2024 Shift 2 - Question 35
Given below are two statements : Statement (I) : A Buffer solution is the mixture of a salt and an acid or a base mixed in any particular quantities. Statement (II) : Blood is naturally occurring buffer solution whose pH is maintained by $$H_2CO_3/HCO_3^-$$ concentrations. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 8th April 2024 Shift 2 - Question 36
The correct sequence of acidic strength of the following aliphatic acids in their decreasing order is: $$CH_3CH_2COOH$$, $$CH_3COOH$$, $$CH_3CH_2CH_2COOH$$, $$HCOOH$$
NTA JEE Mains 8th April 2024 Shift 2 - Question 37
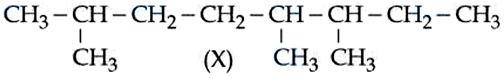
IUPAC name of following hydrocarbon (X) is :
NTA JEE Mains 8th April 2024 Shift 2 - Question 38
Given below are two statements : Statement (I) : Kjeldahl method is applicable to estimate nitrogen in pyridine. Statement (II) : The nitrogen present in pyridine can easily be converted into ammonium sulphate in Kjeldahl method. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 8th April 2024 Shift 2 - Question 39
In qualitative test for identification of presence of phosphorous, the compound is heated with an oxidising agent. Which is further treated with nitric acid and ammonium molybdate respectively. The yellow coloured precipitate obtained is :
NTA JEE Mains 8th April 2024 Shift 2 - Question 40
The emf of cell
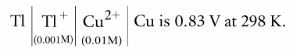
It could be increased by :


