NTA JEE Mains 4th April Shift 2 2026
For the following questions answer them individually
NTA JEE Mains 4th April Shift 2 2026 - Question 61
Consider the following molecules/species:
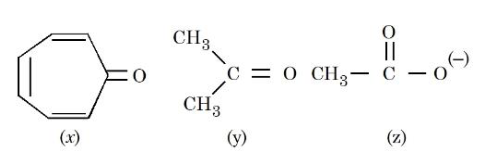
The correct order of carbon-oxygen double bond length is:
NTA JEE Mains 4th April Shift 2 2026 - Question 62
Consider $$|x|$$ is the difference in oxidation states of Mn in highest manganese fluoride and highest manganese oxide. The ions with $$|x|$$ number of unpaired electrons from the following are:
A. Sc$$^{3+}$$
B. Zn$$^{2+}$$
C. V$$^{2+}$$
D. Fe$$^{2+}$$
E. Co$$^{2+}$$
choose the correct answer from the options given below :
NTA JEE Mains 4th April Shift 2 2026 - Question 63
Consider the given graph showing variation of reactant concentration with time. Three different reactions were started with identical initial concentration of reactants. Which of the following statement is correct?
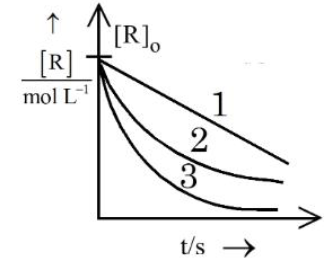
NTA JEE Mains 4th April Shift 2 2026 - Question 64
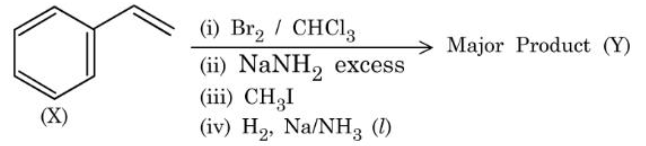
Compound (X) is subjected to the sequence of reactions as shown above:
Molar mass of the major product (Y) formed is ______ g mol$$^{-1}$$.
(Given molar mass in g mol$$^{-1}$$ C:12, H: 1, O: 16)
NTA JEE Mains 4th April Shift 2 2026 - Question 65
The following structures are
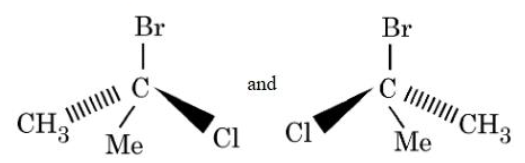
NTA JEE Mains 4th April Shift 2 2026 - Question 66
The descending order of acidity among the following compounds is:
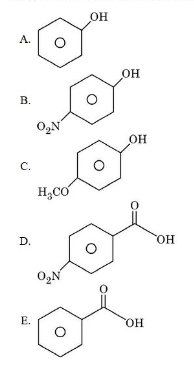
choose the correct answer from the options given below:
NTA JEE Mains 4th April Shift 2 2026 - Question 67
The strongest conjugate acid will result from:
NTA JEE Mains 4th April Shift 2 2026 - Question 68
A D-aldotetrose on oxidation with concentrated HNO$$_3$$ resulted in optically inactive dicarboxylic acid. The structure of the D-aldotetrose is:
NTA JEE Mains 4th April Shift 2 2026 - Question 69
Among Fe$$^{3+}$$, Pb$$^{2+}$$, Cu$$^{2+}$$ and Mn$$^{2+}$$, identify the one that gets precipitated out while passing H$$_2$$S in presence of NH$$_4$$OH as group reagent. The highest possible oxidation state of the corresponding metal is
NTA JEE Mains 4th April Shift 2 2026 - Question 70
Match the List I with List II:
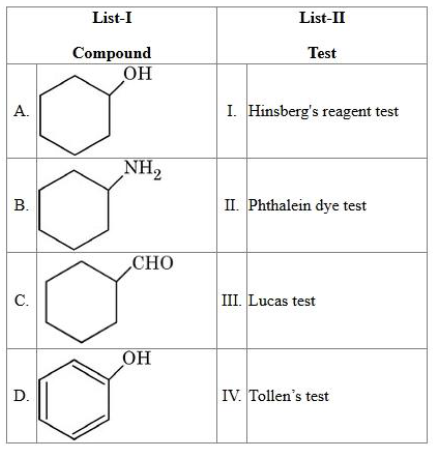
choose the correct answer from the options given below


