NTA JEE Mains 4th April Shift 2 2026
For the following questions answer them individually
NTA JEE Mains 4th April Shift 2 2026 - Question 71
If 3.365 g of ethanol (l) is burnt completely in a bomb calorimeter at 298.15 K, the heat produced is 99.472 kJ. The $$|\Delta H_f°|$$ of ethanol at 298.15 K is _________ $$\times 10^2$$ kJ mol$$^{-1}$$. (Nearest integer)
Given: Standard enthalpy for combustion of graphite = $$-393.5$$ kJ mol$$^{-1}$$
Standard enthalpy of formation of water (l) = $$-285.8$$ kJ mol$$^{-1}$$
Molar mass in g mol$$^{-1}$$ of C, H, O are 12, 1 and 16 respectively
789
456
123
0.-
Clear All
NTA JEE Mains 4th April Shift 2 2026 - Question 72
For the following reaction at 50 °C and at 2 atm pressure,
$$2N_2O_5(g) \rightleftharpoons 2N_2O_4(g) + O_2(g)$$
N$$_2$$O$$_5$$ is 50% dissociated.
The magnitude of standard free energy change at this temperature is $$x$$.
$$x$$ = _________ J mol$$^{-1}$$ [Nearest integer].
Given: $$R = 8.314$$ J mol$$^{-1}$$ K$$^{-1}$$, $$\log 2 = 0.30$$, $$\log 3 = 0.48$$, $$\ln 10 = 2.303$$, °C + 273 = K
789
456
123
0.-
Clear All
NTA JEE Mains 4th April Shift 2 2026 - Question 73
An electrochemical cell, consist of the following two redox couples, M$$^{x+}$$(aq)/M(s) [$$E_{red}^{\Theta} = +0.15$$ V] and Fe$$^{3+}$$(aq)/Fe(s) [$$E_{red}^{\Theta} = -0.036$$ V]. The cell EMF (E$$_{cell}$$) is recorded to be 0.2057 V. If the reaction quotient of the electrochemical reaction is found to be $$10^{-2}$$, then the value of $$x$$ is __________. (Nearest integer)
[Given: M is a p-block metal and $$\frac{2.303RT}{F} = 0.059$$ V]
789
456
123
0.-
Clear All
NTA JEE Mains 4th April Shift 2 2026 - Question 74
For a first order reaction A $$\to$$ B
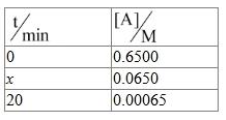
$$x$$ = _________ min. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 4th April Shift 2 2026 - Question 75
In sulphur estimation, $$2.0 \times 10^{-3}$$ mol of an organic compound (X) (molar mass 76 g mol$$^{-1}$$) gave 0.4813 g of barium sulphate (molar mass 233 g mol$$^{-1}$$). The percentage of sulphur in the compound (X) is _________ $$\times 10^{-1}$$ % (Nearest integer)
789
456
123
0.-
Clear All


