NTA JEE Mains 4th April Shift 2 2026
For the following questions answer them individually
NTA JEE Mains 4th April Shift 2 2026 - Question 51
The correct order of total number of atoms in: (A) 2 moles of cyclohexane, (B) 684 g of sucrose, (C) 90.8 L of dihydrogen at STP, is :
NTA JEE Mains 4th April Shift 2 2026 - Question 52
The species having identical radii according to the Bohr's theory are:
A. H (first orbit)
B. He$$^+$$ (first orbit)
C. He$$^+$$ (Second orbit)
D. Li$$^{2+}$$ (first orbit)
E. Be$$^{3+}$$ (Second orbit)
Choose the correct answer from the options given below:
NTA JEE Mains 4th April Shift 2 2026 - Question 53
Which of the following pictorial diagram most correctly represents the $$\pi^*$$ ($$\pi$$ - antibonding) molecular orbital between two atoms if the internuclear axis is taken to be in the z-direction $$(\xrightarrow{z-axis})$$?
NTA JEE Mains 4th April Shift 2 2026 - Question 54
At 27 °C, 0.1 M, 1 L K$$_4$$[Fe(CN)$$_6$$] aqueous solution and 0.1 M, 1 L FeCl$$_3$$ aqueous solution are placed in a container separated by a semi permeable membrane AB. Assume complete dissociation of both the solutes. Which of the following statement is correct?
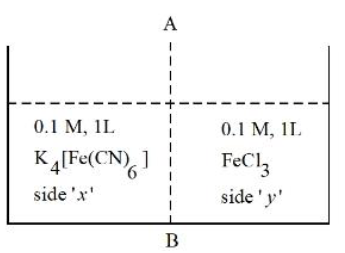
NTA JEE Mains 4th April Shift 2 2026 - Question 55
20 mL of a solution of acetic acid required 28.4 mL of 0.1 M NaOH for its neutralization. A solution (X) was prepared by mixing 20 mL of the above acetic acid and 14.2 mL of 0.1 M NaOH solution. What is the pH of the solution (X)? (pK$$_a$$ value of acetic acid is 4.75).
NTA JEE Mains 4th April Shift 2 2026 - Question 56
Match the List I with List II:
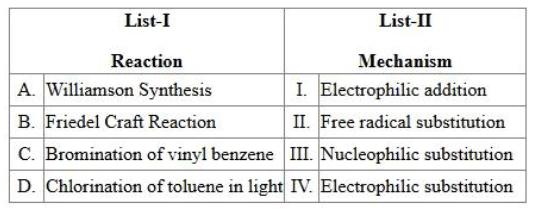
Choose the correct answer from the options given below
NTA JEE Mains 4th April Shift 2 2026 - Question 57
The 1$$^{st}$$ ionization enthalpy for Mg is +737 kJ/mol. The most probable estimated value of the 2$$^{nd}$$ ionization enthalpy of Mg is ______.
NTA JEE Mains 4th April Shift 2 2026 - Question 58
The electronegativity of a group 13 element 'E' is same as that of Ge (on Pauling scale and upto one decimal point). The CORRECT statements about E$$^{3+}$$ are
A. It can act as a reducing agent.
B. It can act as an oxidizing agent.
C. E$$^{3+}$$ is more stable than E$$^+$$.
D. The standard electrode potential value for E$$^{3+}$$/E is positive.
choose the correct answer from the options given below
NTA JEE Mains 4th April Shift 2 2026 - Question 59
Pairs of elements with the same number of electrons in their respective 4f orbital are
[Atomic number. Eu-63, Gd-64, Dy-66, Ho-67, Tm-69, Yb-70, Lu-71, Hf-72]
A. (Eu and Gd)
B. (Dy and Ho)
C. (Yb and Hf)
D. (Lu and Tm)
choose the correct answer from the options given below
NTA JEE Mains 4th April Shift 2 2026 - Question 60
Consider the metal complexes $$[Ni(en)_3]^{2+}$$ (A), $$[NiCl_4]^{2-}$$ (B) and $$[Ni(NH_3)_6]^{2+}$$ (C). Choose the CORRECT option by considering the number of unpaired electrons present in (A), (B) and (C) respectively and the order of frequency of absorption


