NTA JEE Mains 30th Jan 2024 Shift 2
For the following questions answer them individually
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 41
Choose the correct statements about the hydrides of group 15 elements.
A. The stability of the hydrides decreases in the order $$NH_3 > PH_3 > AsH_3 > SbH_3 > BiH_3$$
B. The reducing ability of the hydrides increases in the order $$NH_3 < PH_3 < AsH_3 < SbH_3 < BiH_3$$
C. Among the hydrides, $$NH_3$$ is strong reducing agent while $$BiH_3$$ is mild reducing agent.
D. The basicity of the hydrides increases in the order $$NH_3 < PH_3 < AsH_3 < SbH_3 < BiH_3$$
Choose the most appropriate from the options given below:
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 42
The orange colour of $$K_2Cr_2O_7$$ and purple colour of $$KMnO_4$$ is due to
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 43
$$A$$ and $$B$$ formed in the following reactions are:
$$CrO_2Cl_2 + 4NaOH \rightarrow A + 2NaCl + 2H_2O$$
$$A + 2HCl + 2H_2O_2 \rightarrow B + 3H_2O$$
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 44
Alkaline oxidative fusion of $$MnO_2$$ gives "A" which on electrolytic oxidation in alkaline solution produces $$B$$. $$A$$ and $$B$$ respectively are:
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 45
The molecule/ion with square pyramidal shape is:
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 46
The coordination geometry around the manganese in decacarbonyldimanganese(0) is:
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 47
Given below are two statements:
Statement - I: High concentration of strong nucleophilic reagent with secondary alkyl halides which do not have bulky substituents will follow $$S_N2$$ mechanism.
Statement - II: A secondary alkyl halide when treated with a large excess of ethanol follows $$S_N1$$ mechanism.
In the light of the above statements, choose the most appropriate from the options given below:
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 48
Salicylaldehyde is synthesized from phenol, when reacted with
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 49
m-chlorobenzaldehyde on treatment with 50% KOH solution yields
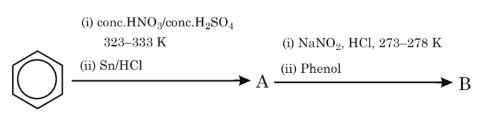
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 50
The products A and B formed in the following reaction scheme are respectively: