NTA JEE Mains 30th Jan 2024 Shift 2
For the following questions answer them individually
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 31
Given below are two statements:
Statement - I: Along the period, the chemical reactivity of the element gradually increases from group 1 to group 18.
Statement - II: The nature of oxides formed by group 1 element is basic while that of group 17 elements is acidic.
In the light of the above statements, choose the most appropriate from the options given below:
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 32
Given below are two statements:
Statement-I: Since fluorine is more electronegative than nitrogen, the net dipole moment of $$NF_3$$ is greater than $$NH_3$$.
Statement-II: In $$NH_3$$, the orbital dipole due to lone pair and the dipole moment of NH bonds are in opposite direction, but in $$NF_3$$ the orbital dipole due to lone pair and dipole moments of N-F bonds are in same direction.
In the light of the above statements, choose the most appropriate from the options given below.
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 33
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: $$H_2Te$$ is more acidic than $$H_2S$$.
Reason R: Bond dissociation enthalpy of $$H_2Te$$ is lower than $$H_2S$$.
In the light of the above statements, choose the most appropriate from the options given below.
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 34
IUPAC name of the following compound is:
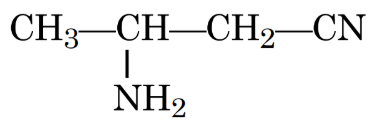
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 35
Which among the following purification methods is based on the principle of "Solubility" in two different solvents?
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 36
The correct stability order of carbocations is
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 37
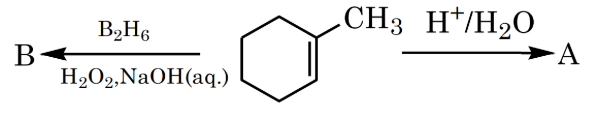
Product A and B formed in the following set of reactions are:
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 38
If a substance '$$A$$' dissolves in solution of a mixture of '$$B$$' and '$$C$$' with their respective number of moles as $$n_A$$, $$n_B$$ and $$n_C$$, mole fraction of $$C$$ in the solution is:
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 39
The solution from the following with highest depression in freezing point/lowest freezing point is
NTA JEE Mains 30th Jan 2024 Shift 2 - Question 40
Reduction potential of ions are given below:
$$ClO_4^-$$: $$E° = 1.19$$ V; $$IO_4^-$$: $$E° = 1.65$$ V; $$BrO_4^-$$: $$E° = 1.74$$ V
The correct order of their oxidising power is:


