NTA JEE Mains 29th Jan 2024 Shift 2
For the following questions answer them individually
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 41
Which of the following statements are correct about Zn, Cd and Hg?
A. They exhibit high enthalpy of atomization as the d-subshell is full.
B. Zn and Cd do not show variable oxidation state while Hg shows +I and +II.
C. Compounds of Zn, Cd and Hg are paramagnetic in nature.
D. Zn, Cd and Hg are called soft metals.
Choose the most appropriate from the options given below:
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 42
The correct IUPAC name of $$K_2MnO_4$$ is:
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 43
Alkyl halide is converted into alkyl isocyanide by reaction with
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 44
Phenol treated with chloroform in presence of sodium hydroxide, which further hydrolysed in presence of an acid results
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 45
Identify the reagents used for the following conversion
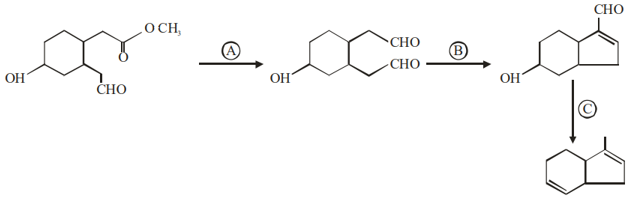
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 46
Which of the following reaction is correct?
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 47
The product A formed in the following reaction is:
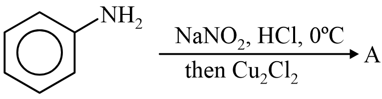
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 48
On passing a gas, '$$X$$', through Nessler's reagent, a brown precipitate is obtained. The gas '$$X$$' is
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 49
A reagent which gives brilliant red precipitate with Nickel ions in basic medium is
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 50
Match List I with List II
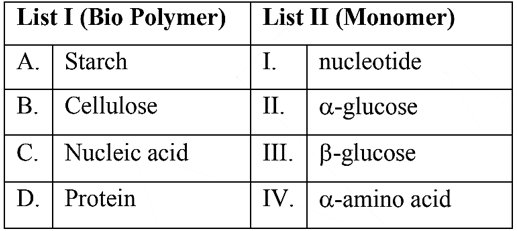
Choose the correct answer from the options given below:


