NTA JEE Mains 29th Jan 2024 Shift 2
For the following questions answer them individually
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 31
Match List I with List II
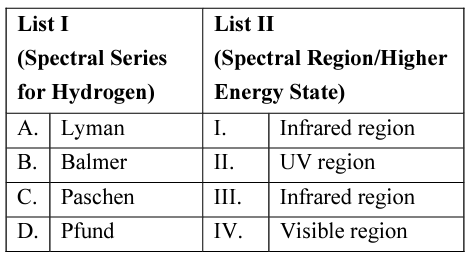
Choose the correct answer from the options given below:
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 32
The element having the highest first ionization enthalpy is
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 33
Given below are two statements:
Statement I: Fluorine has most negative electron gain enthalpy in its group.
Statement II: Oxygen has least negative electron gain enthalpy in its group.
In the light of the above statements, choose the most appropriate from the options given below.
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 34
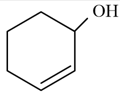
According to IUPAC system, the compound is named as:
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 35
The ascending acidity order of the following H atoms is
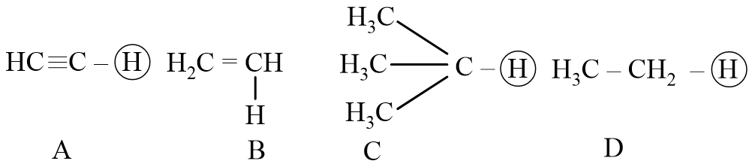
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 36
Match List I with List II
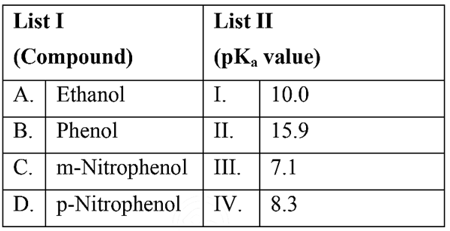
Choose the correct answer from the options given below:
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 37
Which one of the following will show geometrical isomerism?
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 38
Chromatographic technique/s based on the principle of differential adsorption is/are
A. Column chromatography B. Thin layer chromatography C. Paper chromatography
Choose the most appropriate answer from the options given below:
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 39
Anomalous behaviour of oxygen is due to its
NTA JEE Mains 29th Jan 2024 Shift 2 - Question 40
Which of the following acts as a strong reducing agent? (Atomic number: Ce = 58, Eu = 63, Gd = 64, Lu = 71)


