NTA JEE Mains 28th Jan 2026 Shift 2
For the following questions answer them individually
NTA JEE Mains 28th Jan 2026 Shift 2 - Question 61
Match List - I with List - Il according to shape.
List - I List - II
A. $$ XeO_3 $$ I. $$ BrF_5 $$
B. $$ XeF_2 $$ II. $$ NH_3 $$
C. $$ XeO_2 F_2 $$ III. $$ [I_3]^-{} $$
D. $$ XeOF_4 $$ IV. $$ SF_4 $$
Choose the correct answer from the options given below:
NTA JEE Mains 28th Jan 2026 Shift 2 - Question 62
The plot of $$\log_{10}^{K} VS \frac{1}{T}$$ gives a straight Line. The intercept and slope respectively are
(where K is equilibrium constant).
NTA JEE Mains 28th Jan 2026 Shift 2 - Question 63
Consider the elements N, P, 0, S,Cl and F. The number of valence electrons present in the elements with most and least metallic character from the above list is respectively.
NTA JEE Mains 28th Jan 2026 Shift 2 - Question 64
Consider the following aqueous solutions.
I. 2.2 g Glucose in 125 ml of solution.
II. 1.9 g Calcium chloride in 250 ml of solution.
111. 9.0 g Urea in 500 ml of solution.
IV. 20.5 g Aluminium sulphate in 750 ml of solution.
The correct increasing order of boiling point of these solutions will be:
[Given : Molar mass in g $$mol^{-1}$$: H = 1, C=12, N= 14, 0=16, Cl =35.5, Ca=40, Al=27 and S=32]
NTA JEE Mains 28th Jan 2026 Shift 2 - Question 65
Consider the following reactions
$$\begin{aligned}\mathrm{Na_2B_4O_7} & \xrightarrow{\Delta} 2X + Y \\[6pt]\mathrm{CuSO_4} + Y &\xrightarrow{\text{Non-luminous flame}} Z + \mathrm{SO_3}\\[6pt]2Z + 2X + \mathrm{C}&\xrightarrow{\text{Luminous flame}} 2Q +\mathrm{Na_2B_4O_7} + \mathrm{CO}\end{aligned}$$
The oxidation states of Cu in Z and Q, respectively are:
NTA JEE Mains 28th Jan 2026 Shift 2 - Question 66
Consider the following statements about manganate and permanganate ions. Identify the correct statements.
A. The geometry of both manganate and permanganate ions is tetrahedral
B. The oxidation states of Mn in manganate and permanganate are + 7 and + 6, respectively.
C. Oxidation of Mn(II) salt by peroxodisulphate gives manganate ion as the final product.
D. Manganate ion is paramagnetic and permanganate ions is diamagnetic.
E. Acidified permanganate ion reduces oxalate, nitrite and iodide ions.
Choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2026 Shift 2 - Question 67
A student has been given 0.314 g of an organic compound and asked to estimate Sulphur. During the experiment, the student has obtained 0.4813 g of barium sulphate. The percentage of sulphur present in the compound is_______.{Given Molor mass in g $$mol^{-1}$$ S: 32, $$BaSO_{4}$$ : 233)
NTA JEE Mains 28th Jan 2026 Shift 2 - Question 68
The cyclic cations having the same number of hyperconjugalion are :
A.
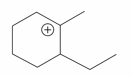
B.
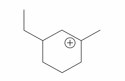
C.
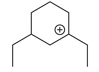
D.
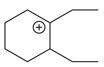
Choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2026 Shift 2 - Question 69
The correct order of acidic strength of the major products formed in the given reactions, is:
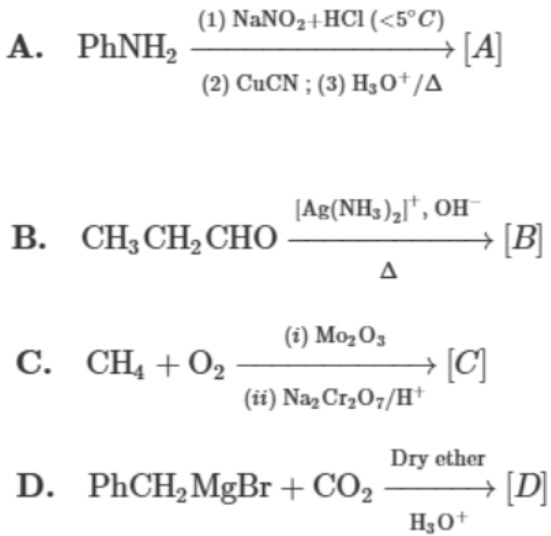
Choose the correct answer from the options given below :
NTA JEE Mains 28th Jan 2026 Shift 2 - Question 70
The reactions which produce alcohol as the product are:
$$A. CH_{4}+O_{2}\xrightarrow[\Delta]{Mo_{2}O_{3}}\\B.2CH_{3}CH_{3}+3O_{2}\xrightarrow[\Delta]{(CH_{3}COO)_{2}Mn}\\C.(CH_{3})_{3}CH\xrightarrow{KMnO_{4}}\\D.2CH_{4}+O_{2}\xrightarrow{Cu/523K/100atm.}\\E.CH_{3}-CH=CH-CH_{3}\xrightarrow{KMnO_{4}/H^{+}}$$
Choose the correct answer from the options given below :


