NTA JEE Mains 28th Jan 2026 Shift 2
For the following questions answer them individually
NTA JEE Mains 28th Jan 2026 Shift 2 - Question 71
A volume of x mL of 5 M $$NaHCO_{3}$$ solution was mixed with 10 mL of 2 M $$H_{2}CO_{3}$$ solution to make an electrolytic buffer. If the same buffer was used in the following electrochemical cell to record a cell potential of 235.3 mV, then the value of x=_______ mL (nearest integer).
$$Sn(s)|Sn(OH)_{6}^{2-}(0.5 M)|HSnO_{2}^{-}(0.05 M)|OH^{-}|Bi_{2}O_{3}(s)|Bi(s)$$
Consider upto one place of decimal for intermediate calculations

789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2026 Shift 2 - Question 72
The numbei of isoelectronic species among $$Sc^{3+},Cr^{2+},Mn^{3+},Co^{3+}\text{ and }Fe^{3+}$$ is 'n'. If 'n' moles of AgCl is formed during the reaction of complex with formula $$CoCl_{3}(en)_{2}NH_{3}$$ with excess of $$AgNO_{3}$$ solution, then the number of electrons present in the $$t_{2g}$$ orbital of the complex is ________.
789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2026 Shift 2 - Question 73
For strong electrolyte $$\lambda_m$$ increases slowly with dilution and can be represented by the equation
$$\Lambda_m = \Lambda_m^\circ - A c^{1/2}$$
Molar conductivity values of the solutions of strong electrolyte AB at 18°C are given below:
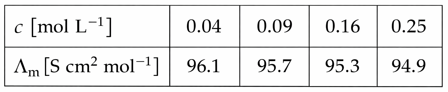
The value of constant A based on the above data [in S $$cm^{2}mol^{-1}/(mol/L)^{1/2}$$]unit is_______.
789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2026 Shift 2 - Question 74
Two positively charged particles m1 and m2 have been accelerated across the same potential difference of 200 keV as shown below.
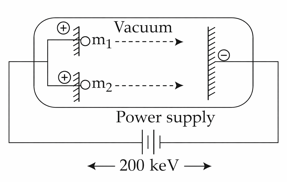
[Given mass of $$m_{1}$$ = 1amu and $$m_{2}$$ = 4amu]
The deBroglie wavelength of $$m_{1}$$ will be x times of $$m_{2}$$. The value of x is_______(nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 28th Jan 2026 Shift 2 - Question 75
$$A \rightarrow B$$ (first reaction)
$$C \rightarrow D$$ (second reaction)
Consider the above two first-order reactions. The rate constant for first reaction at 500 K is double of the same at 300 K. At 500 K, 50% of the reaction becomes complete in 2 hour. The activation energy of the second reaction is half of that of first reaction. lf the rate constant at 500 K of the second reaction becomes double of the rate constant of first reaction at the same temperature; then rate constant for the second reaction at 300 K is______$$\times 10^{-1}hour^{-1}$$ (nearest integer).
789
456
123
0.-
Clear All


