NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 51
Given below are h¥o statements:
Statement I: Hybridisation, shape and spin only magnetic moment of $$K_{3}[Co(CO_{3})_{3}]$$ is $$sp^{3}d^{2}$$, octahedral and 4.9 BM respectively.
Statement II: Geometty, hybridisation and spin only magnetic moment values (BM) of the ions $$[Ni(CN)_{4}]^{2-},[MnBr_{4}]^{2-}\text{ and }[CoF_{6}]^{3-}$$ respectively are square planar, tetrahedral, octahedral;$$dsp^{2},sp^{3},sp^{3}d^{2}$$ and 0, 5.9, 4.9.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 52
Arrange the following carbanions in the decreasing order of stability.
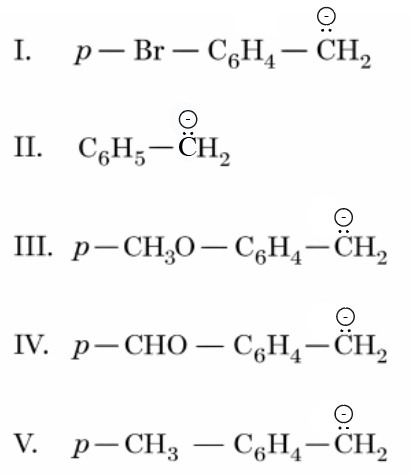
Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 53
Consider three metal chlorides x, y and z, where x is water soluble at room temperature, y is sparingly soluble in water at room temperature and z is soluble in hot water. x, y and z are respectively
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 54
At 27 °C in presence of a catalyst, activation energy of a reaction is lowered by $$10 \text{KJ mol}^{-1}$$. The logarithm of ratio of $$\frac{k(\text{catalysed})}{k(\text{uncatalysed})}$$ is......
(Consider that the frequency factor for both the reactions is same)
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 55
A hydroxy compound (X) with molar mass 122 g $$mol^{-1}$$ is acetylated with acetic anhydride, using a large excess of the reagent ensuring complete acetylation of all hydroxyl groups. The product obtained has a molar mass of 290 g $$mol^{-1}$$. The number of hydroxyl groups present in compound (X) is:
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 56
Anange the following alkenes in decreasing order of stability.
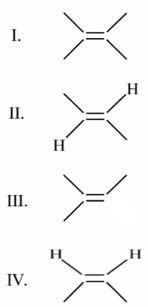
Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 57
Given below are two statements:
Statement I: 'C - Cl' bond is stronger in $$CH_{2}=CH-Cl\text{ and }CH_{3}-CH_{2}-Cl$$
Statement II: The given optically active molecule,
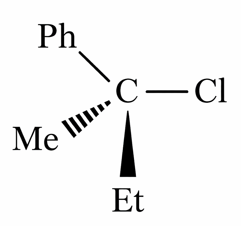
on hydrolysis gives a solution that can rotate the plane polarized light.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 58
Consider a mixtm-e 'X' which is made by dissolving 0.4 mol of $$[Co(NH_{3})_{5}SO_{4}]Br$$ and 0.4 mol of $$[Co(NH_{3})_{5}Br]SO_{4}$$ in water to make 4 L of solution. When 2 L of mixture 'X' is allowed to react with excess of $$AgNO_{3}$$,it fonns precipitate 'Y'. The rest 2 L of mixture 'X' reacts with excess $$BaCl_{2}$$ to fonn precipitate 'Z'. Which of the following statements is CORRECT?
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 59
Given below are two statements:
Statement I: K > Mg > Al > B is the correct order in terms of metallic character.
Statement II: Atomic radius is always greater than the ionic radius for any element.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 60
Given below are statements about some molecules/ions.
Identify the CORRECT statements.
A The dipole moment value of $$NF_{3}$$ is higher than that of $$NH_{3}$$.
B. The dipole moment value of $$BeH_{2}$$ is zero.
C. The bond order of $$O_{2}^{2-}$$ and $$F_{2}$$ is same.
D. The formal charge on the central oxygen atom of ozone is -1 .
E. In $$NO_{2}$$, all the three atoms satisfy the octet rule, hence it is very stable.
Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 61
A solution is prepared by dissolving 0.3 g of a non-volatile non-electrolyte solute 'A' of molar mass 60 g $$mol^{-1}$$ and 0.9 g of a non-volatile non-electrolyte solute 'B' of molar mass 180 g $$mol^{-1}$$ in 100 mL $$H_{2}O$$ at 27°C. Osmotic pressure of the solution will be
[Given: R = 0.082 L atm $$K^{-1} mol^{-1}$$]
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 62
Given below are two statements:
Statement I: The number of paramagnetic species among $$[CoF_{6}]^{3-},[TiF_{6}]^{3-},V_{2}O_{5}\text{ and }[Fe(CN)_{6}]^{3-}$$ is 3.
Statement II: $$K_{4}[Fe(CN)_{6}] < K_{3}[Fe(CN)_{6}] < [Fe(H_{2}O)_{6}]SO_{4}.H_{2}O < [Fe(H_{2}O)_{6}]Cl_{3}$$ is the correct order in terms of number of unpaired electron(s) present in the complexes.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 63
Among the following, the CORRECT combinations are
$$\text{A. IF}_{3} \rightarrow \text{T-shaped}(\text{sp}^{3}\text{d})$$
$$\text{B. IF}_{5} \rightarrow \text{Square pyramidal(sp}^{3}\text{d}^{2})$$
$$\text{C. IF}_{7} \rightarrow \text{Pentagonal bipyrnmidal(sp}_{3}\text{d}^{3})$$
$$\text{D. ClO}_{4}^{-} \rightarrow \text{Square planar(sp}^{3}\text{d})$$
Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 64
'W' g of a non-volatile electrolyte solid solute of molar mass 'M' g $$mol^{-1}$$ when dissolved in 100 mL water, decreases vapour pressure of water from 640 mm Hg to 600 mm Hg. If aqueous solution of the electrolyte boils at 375 K and $$K_{b}$$ for water is 0.52 K kg $$mol^{-1}$$, then the mole fraction of the electrolyte solute ($$x_{2}$$) in the solution can be expressed as
(Given : density of water= 1 g/mL and boiling point of water= 3 73 K)
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 65
Consider the following two reactions A and B.
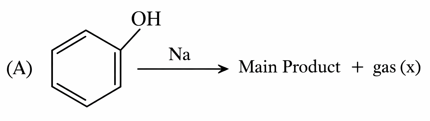
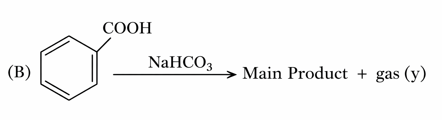
Numerical value of [ molar mass of x + molar mass of y] is __
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 66
Match the LIST-I with LIST-II
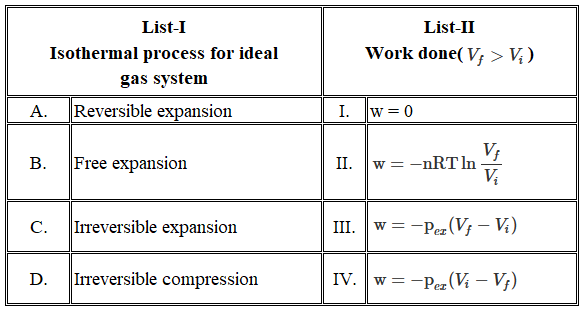
Choose the correct answer from the options given befow:
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 67
A student is given one compound among the following compounds that gives positive test with Tollen's reagent
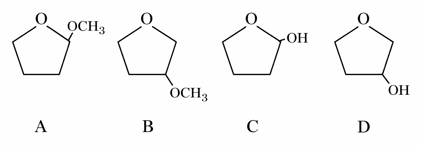
The compound is :
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 68
The correct stability order of the following diazonium salts is
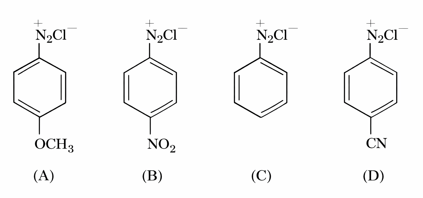
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 69
$$A\rightarrow D$$ is an endothermic reaction occurring in three steps ( elementary).
(i) $$A\rightarrow B \triangle H_{i}=+ve$$
(ii) $$B\rightarrow C \triangle H_{ii}=-ve$$
(iii) $$C\rightarrow D \triangle H_{iii}=-ve$$
Which of the following graphs between potential energy (y-axis) vs reaction coordinate (x-axis) correctly represents the reaction profile of A-> D?
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 70
Match the LIST-I with LIST-II
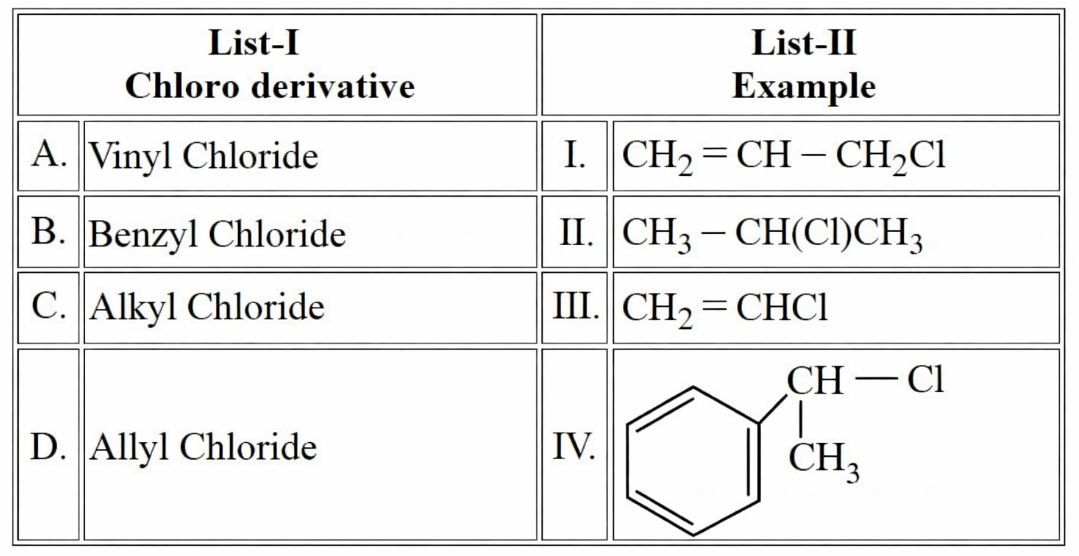
Choose the correct answer from the options given below:
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 71
Consider two Group IV metal ious $$X^{2+}\text{ and }Y^{2+}$$
A solution containing $$0.01 M X^{2+}\text{ and }0.01MY^{2+}$$ is satmated with $$H_{2}S$$. The pH at which the metal sulphide YS will form as a precipitate is __ . (Nearest integer)
$$(Given:K_{sp}(XS)=1\times 10^{-22} \text{ at } 25^{\circ}C,K_{sp}(YS)=4\times 10^{-16} \text{ at } 25^{\circ}C,[H_{2}S]=0.1M\text{ in solution },K_{a1}\times K_{a2}(H_{2}S)=1.0\times 10^{-21},\log{2}=0.30,\log{3}=0.48,\log{5}=0.70)$$
789
456
123
0.-
Clear All
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 72
In Dumas method for estimation of nitrogen, 0.50 g of an organic compound gave 70 mL of nitrogen collected at 300 K and 715 mm pressure. The percentage of nitrogen in the organic compound is __ %.
(Aqueous tension at 300 K is 15 mm).
789
456
123
0.-
Clear All
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 73
X and Y are the number of electrons involved, respectively during the oxidation of $$\ I^{-}\text{ to }I_{2}\text{ and }S^{2-}$$ to S by acidified $$K_{2}Cr_{2}O_{7}$$. The value of X + Y is __ .
789
456
123
0.-
Clear All
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 74
Electricity is passed through an acidic solution of $$Cu^{2+}$$ till all the Cu2+ was exhausted, leading to the deposition of 300 mg of Cu metal. However, a current of 600 mA was continued to pass through the same solution for another 28 minutes by keeping the total volume of the solution fixed at 200 mL. The total volume of oxygen evolved at STP during the entire process is __ mL. (Nearest integer)
[Given:
$$Cu^{2+}(aq)+2e^{-}\rightarrow Cu(s) E_{red}^{\circ}=+0.34V$$
$$O_{2}(g)+4H^{+}+4e^{-}\rightarrow 2H_{2}O E_{red}^{\circ}=+1.23V$$
Molar mass of Cu= 63.54 g $$mol^{-1}$$
Molar mass of $$O_{2}$$ = 32 g $$mol^{-1}$$
Farnday Constant = 96500 C $$mol^{-1}$$
Molar volume at STP = 22.4 L]
789
456
123
0.-
Clear All
NTA JEE Mains 24th Jan 2026 Shift 1 - Chemistry - Question 75
The hydrogen spectnun consists of several spectral lines in Lyman series ($$L_{1},L_{2},L_{3}....;L_{1}$$ has lowest energy among Lyman series). Similarly it consists of several spectral lines in Balmer series($$B_{1},B_{2},B_{3}....;B_{1}$$ has lowest energy among Balmer lines). The energy of $$L_{1}$$ is x times the energy of $$B_{1}$$. The value of x is_____ $$\times 10^{-1}$$. (Nearest integer)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




