NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 51
Match List - I with List - II.
.png)
choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 52
The statements that are incorrect about the nickel(II) complex of dimethylglyoxime are:
A. It is red in colour.
B. It has a high solubility in water at pH =9.
C. The Ni ion has two unpaired d-electrons.
D. The N - Ni - N bond angle is almost close to 90°
E. The complex contains four five-membered metallacycles (metal containing rings).
Choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 53
The correct statements from the following are:
A. Ionic radii of trivalent cations of group 13 elements decreases down the group.
B. Electronegativity of group 13 elements decreases down the group.
C. Among the group 13 elements, Boron has highest first ionisation enthalpy.
D. The trichloride and triiodide of group 13 elements are covalent in nature.
Choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 54
Consider the following compounds
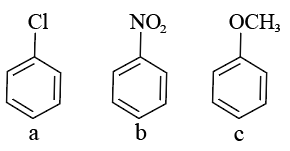
Arrange these compounds in the increasing order of reactivity with nitrating mixture.
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 55
Given below are two statements :
Statement I: $$[CoBr_{4}]^{2-}$$ ion will absorb light of lower energy than $$[CoCl_{4}]^{2-}$$ ion.
Statement II: In $$[Col_{4}]^{2-}$$ ion, the energy separation between the two set of d-orbitals is more than $$[CoCl_{4}]^{2-}$$ ion.
ln the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 56
Which one of the following graphs accurately represents the plot of partial pressure of $$CS_{2}$$ vs its mole fraction in a mixture of acetone and $$CS_{2}$$ at constant temperature?
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 57
Given below are two statements :
Statement I: Sublimation is used for the separation and purification of compounds with low melting Point.
Statement II: The boiling point of a liquid increases as the external pressure is reduced.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 58
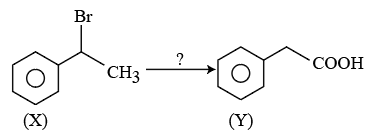
The correct sequence of reagents for the above conversion of X to Y is:
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 59
Consider the following sequence of reactions.
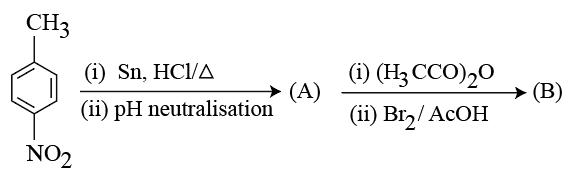
4-Nitrotoluene
Assuming that the reaction proceeds to completion, then 137 mg of 4-nitrotoluene will produce ____ mg of B.
(Given molar mass in g $$mol^{-1}$$ H : 1, C: 12, N: 14, 0: 16, Br : 80)
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 60
The correct trend in the first ionization enthalpies of the elements in the $$3^{rd}$$ period of periodic table is:
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 61
Which of the following statements regarding the energy of the stationary state is true in the following one - electron systems?
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 62
Consider the general reaction given below at 400 K
$$xA(g)\rightleftharpoons yB(g).
The values of $$K_{p}\text{ and }K_{c}$$ are studied under the same condition of temperature but variation in x and y
(i)$$K_{p}=85.87\text{ and }K_{c}=2.586$$ appropriate units
(ii)$$K_{p}=0.862\text{ and }K_{c}=28.62$$ appropriate units
The values of x and yin (i) and (ii) respectively are:
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 63
Compound 'P' undergoes the following sequence of reactions:
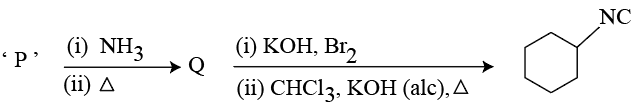
'P' is:
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 64
A cup of water at 5°C (system) is placed in a microwave oven and the oven is turned on for one minute during which the water begins to boil. Which of the following option is true ?
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 65
ldentify the molecule (X) with maximum number of lone pairs of electrons (obtained using Lewis dot structure) among $$HNO_{3},H{2}SO_{4},NF_{3}\text{ and }O_{3}$$. Choose the correct bond angle made by the central atom of the molecule (X).
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 66
From the given following (A to D) cyclic structures, those which will not react with Tollen's reagent are:
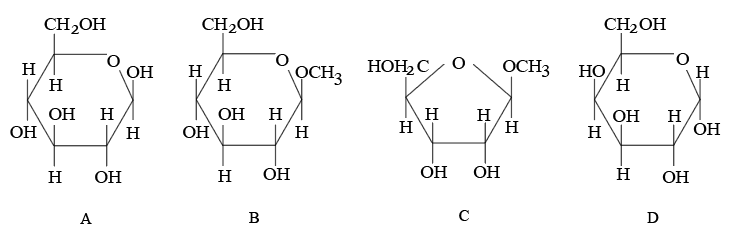
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 67
But-2-yne and hydrogen (one mole each) are separately treated with (i) Pd/C and (ii) Na/ liq. NH3 to give the products X and Y respectively.
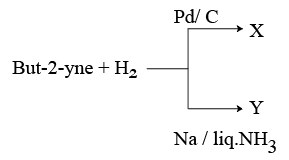
Identify the incorrect statements.
A. X and Y are stereoisomers.
B. Dipole moment of X is zero.
C. Boiling point of X is higher than Y.
D. X and Y react with $$O_{3}/Zn+H_{2}O$$ to give different products.
Choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 68
'x' is the product which is obtained &om propanenitrile and stannous chloride in the presence of hydrochloric acid followed by hydrolysis. 'y' is the product which is obtained from the but-2-ene by the ozonolysis followed by hydrolysis. From the followu1g, which product is not obtained when one mole of 'x' and one mole of 'y' react with, each other in the presence of alkali followed by heating?
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 69
In the given electrochemical cell, $$Ag(s)|AgCl(s)|FeCl_{2}(aq)$$, $$FeCl_{3}(aq)|Pt(s)$$ at298 K, theceU potential $$E_{cell}$$ will increase when:
A. Concentration of $$Fe^{2+}$$ is increased.
B. Concentration of $$Fe^{3+}$$ is decreased.
C. Concentration of $$Fe^{2+}$$ is decreased.
D. Concentration of $$Fe^{3+}$$ is increased.
E. Concentration of $$Cl^{-}$$ is increased.
Choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 70
Given,
(A) $$n = 5, m_{1} = -1$$
(B) $$n = 3, 1 = 2, m_{1} = -1, m_{2} = +\frac{1}{2}$$
The maximum number of electron(s) in an atom that can have the quantum numbers as given in (A) and (B) respectively are:
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 71
The crystal field splitting energy of $$[Co(oxalate)_{3}]^{3-}$$ complex is 'n' times that of the $$[Cr(oxalate)_{3}]^{3-}$$ complex. Here 'n' is_______. (Assume d $$\triangle_{\circ} > > P$$)
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 72
For the following gas phase equilibrium reaction at constant temperature,
$$NH_{3}(g)\rightleftharpoons 1/2N_{2}(g)+3/2H_{2}(g)$$
if the to tal pressure is $$\sqrt{3}$$ atm and the pressure equilibrium constant ($$K_{p}$$) is 9 atm, then the degree of dissociation is given as $$(x\times 10^{-2})^{-1/2}$$.The value of x is ______. (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 73
x mg of pure HCl was used to make an aqueous solution. 25.0 mL of 0.1 M $$Ba(OH)_{2}$$ solution is used when the HCl solution was titrated against it. The numerical value of x is ______$$\times 10^{-1}$$. (nearest integer)
Given : Molar mass of HCl and $$Ba(OH)_{2}$$ are 36.5 and 171.0 g $$mol^{-1}$$ respectively.
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 74
Consider all the structural isomers with molecular formula $$C_{5}H_{11}Br$$ are separately treated with KOH(aq) to give respective substitution products, without any rearrangement. The number of products which can exhibit optical isomerism from these is ______.
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2026 Shift 1 - Chemistry - Question 75
For the thermal decomposition of reactant AB(g), the following plot is constructed.
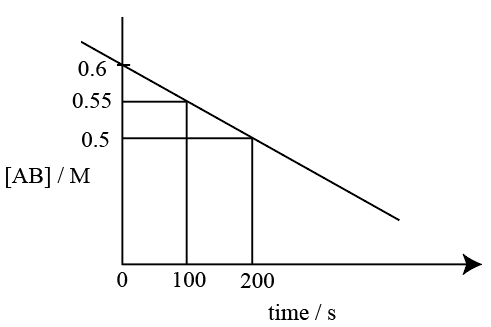
The half life of the reaction is 'x' min.
x= ____ min. (Nearest integer)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




