NTA JEE Mains 23rd Jan 2025 Shift 2
For the following questions answer them individually
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 61
Given below are two statements about X-ray spectra of elements : Statement (I) : A plot of $$\sqrt{v}$$($$\upsilon$$ = frequency of -rays emitted) vs atomic mass is a straight line. Statement (II) : A plot of $$\upsilon(\upsilon$$ = frequency of -rays emitted) vs atomic number is a straight line. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 62
Identify A,B and C in the given below reaction sequence
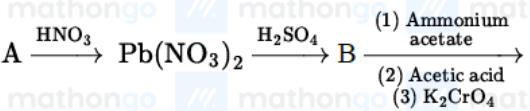
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 63
Given below are two statements : Statement (I) : The boiling points of alcohols and phenols increase with increase in the number of C-atoms. Statement (II) : The boiling points of alcohols and phenols are higher in comparison to other class of compounds such as ethers, haloalkanes. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 64
Consider a binary solution of two volatile liquid components 1 and 2.$$x_{1}$$ and $$y_{1}$$ are the mole fractions of component 1 in liquid and vapour phase, respectively. The slope and intercept of the linear plot of $$\frac{1}{x_{1}}$$ vs $$\frac{1}{y_{1}}$$ are given respectively as:
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 65
The ascending order of relative rate of solvolysis of following compounds is :
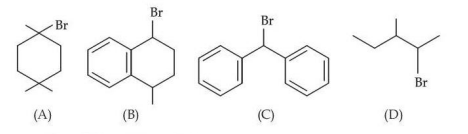
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 66
Match List - I with List - II.

Choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 67
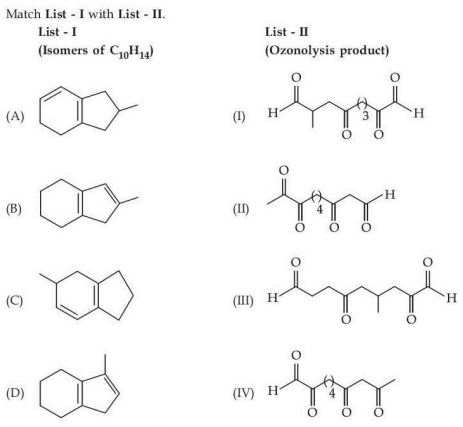
Choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 68
pH of water is 7 at $$25^{\circ}C$$.If water is heated to $$80^{\circ}C$$.,it's pH will :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 69
Identify the coordination complexes in which the central metal ion has $$d^{4}$$ configuration.
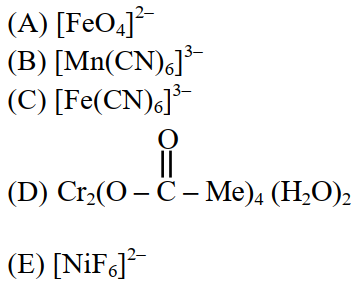
Choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 70
When a non-volatile solute is added to the solvent, the vapour pressure of the solvent decreases by 10 mm of Hg . The mole fraction of the solute in the solution is 0.2 . What would be the mole fraction of the solvent if decrease in vapour pressure is 20 mm of Hg ?


