NTA JEE Mains 23rd Jan 2025 Shift 2
For the following questions answer them individually
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 71
0.01 mole of an organic compound (X) containing 10% hydrogen, on complete combustion produced $$0.9_{g}H_{2}O$$. Molar mass of (X) is_____$$mol^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 72
A compound 'X' absorbs 2 moles of hydrogen and 'X' upon oxidation with $$KMnO_{4}|H^{+}$$ gives

The total number of $$\sigma$$ bonds present in the compound 'X' is ________.
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 73
When 81.0 g of aluminium is allowed to react with 128.0 g of oxygen gas, the mass of aluminium oxide produced in grams is_______ - (Nearest integer) Given : Molar mass of Al is 27.0 g $$mol^{-1}$$ Molar mass of O is 16.0 g $$mol^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 74
The bond dissociation enthalpy of $$X_{2}\Delta H_{bond}$$ calculated from the given data is_____$$kJmol^{-1}$$.(Nearest integer)
$$M^{+}X^{-}(s)\rightarrow M^{+}(g)+X^{-}(g)\Delta H_{lattice}^{*}=800kJmol^{-1}\\M(s)\rightarrow M(g)\Delta H_{sub}^{\circ}=100kJmol^{-1}\\M(g)\rightarrow M^{+}(g)+e^{-}(g)\Delta H_{i}=500kJmol^{-1}X(g)+e^{-}(g)\rightarrow X^{-}(g)\Delta H_{eg}^{*}=-300kJmol^{-1}\\M(g)+\frac{1}{2}X_{2}(g)\rightarrow M^{+}X^{-}(s)\Delta H_{f}^{\circ}=-400kJmol^{-1}$$
[Given : $$M^{+}X^{-}$$ is a pure ionic compound and X forms a diatomic molecule $$X_{2}$$ in gaseous state]
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 75
Consider the following sequence of reactions.
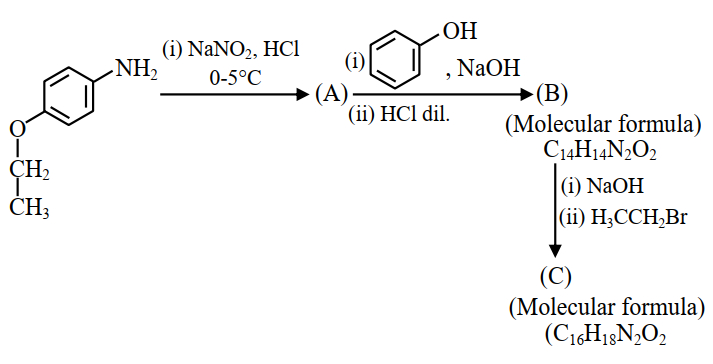
Total number of $$sp^{3}$$ hybridised carbon atoms in the major product C formed is________
789
456
123
0.-
Clear All


