NTA JEE Mains 23rd Jan 2025 Shift 2
For the following questions answer them individually
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 51
Identify the products [A] and [B], respectively in the following reaction :
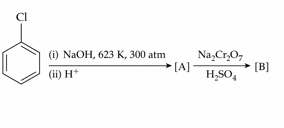
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 52
Consider the following reactions $$K_{2}Cr_{2}O_{7}\xrightarrow[-H_{2}O]{KOH}[A]\xrightarrow[-H_{2}O]{H_{2}SO_{4}}[B]+K_{2}SO_{4}$$ The products [A] and [B], respectively are :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 53
The effect of temperature on spontaneity of reactions are represented as :

NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 54
Which of the following graphs most appropriately represents a zero order reaction ?
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 55
Consider the reaction $$X_{2}Y(g)=X_{2}(g)+\frac{1}{2}Y_{2}(g)$$ The equation representing correct relationship between the degree of dissociation (x) of $$X_{2}Y(g)$$ with its equilibrium constant Kp is ______ . Assume x to be very very small.
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 56
Given below are two statements : Consider the following reaction
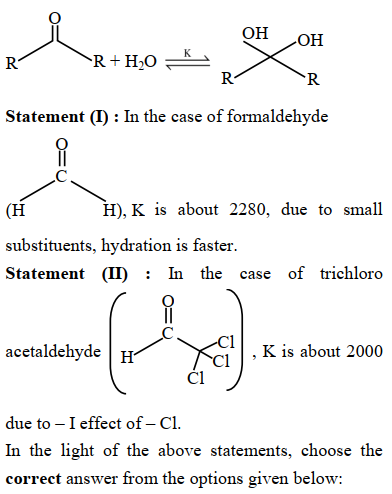
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 57
Given below are two statements : Statement (I) : For a given shell, the total number of allowed orbitals is given by $$n^{2}$$. Statement (II) : For any subshell, the spatial orientation of the orbitals is given by -l to +l values including zero. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 58
Standard electrode potentials for a few half cells are mentioned below :
$$E_{Cu^{2+}/Cu}^{\circ}=0.34 V,E_{Zn^{2+}/Zn}^{\circ}=-0.76 V\\E_{Ag^{+}/Ag}^{\circ}=0.80 V,E_{Mg^{2+}/Mg}^{\circ}=-2.37 V$$ Which one of the following cells gives the most negative value of $$\Delta G^{\circ}$$ ?
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 59
The $$\alpha$$-Helix and $$\beta$$- Pleated sheet structure of protein are associated with its :
NTA JEE Mains 23rd Jan 2025 Shift 2 - Question 60
Given below are the atomic numbers of some group 14 elements. The atomic number of the element with lowest melting point is :


