NTA JEE Mains 23rd Jan 2025 Shift 1
For the following questions answer them individually
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 61
$$CrCl_{3}\cdot xNH_{3}$$ can exist as a complex. 0.1 molal aqueous solution of this complex shows a depression in freezing point of $$0.558^{\circ}C$$. Assuming 100% ionisation of this complex and coordination number of Cr is 6 , the complex will be (Given $$K_{f}$$ = 1.86 K kg $$mol^{-1}$$)
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 62
Which of the following happens when $$NH_{4}OH$$ is added gradually to the solution containing 1 M $$A^{2+}$$ and $$1MB^{3+}$$ ions? Given : $$K_{sp}[A(OH)_{2}]= 9 \times 10^{-10}$$ and $$K_{sp}[B(OH)_{3}]= 27 \times 10^{-18}$$ at 298 K .
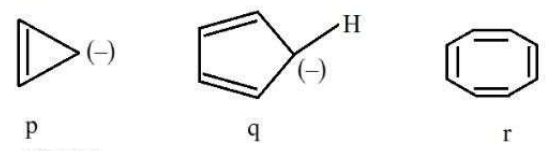
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 63
The major product of the following reaction is:
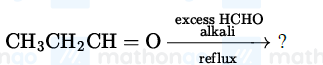
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 64
Ice at $$-5^{\circ}C$$ is heated to become vapor with temperature of $$110^{\circ}C$$ at atmospheric pressure. The entropy change associated with this process can be obtained from
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 65
The incorrect statement among the following is options .
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 66
$$2.8 \times 10^{-3}$$ mol of $$CO_{2}$$ is left after removing $$10^{21}$$ molecules from its 'x' mg sample. The mass of $$CO_{2}$$ taken initially is Given: $$N_{A} = 6.02 \times 10^{23} mol^{-1}$$
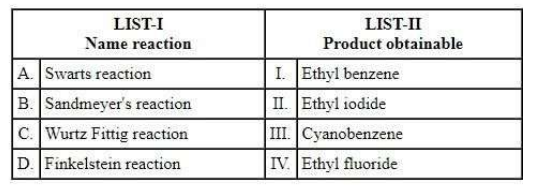
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 67
Match the LIST-I with LIST-II
Choose the correct answer from the options given below:
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 68
Heat treatment of muscular pain involves radiation of wavelength of about 900 nm . Which spectral line of H atom is suitable for this? Given : Rydberg constant $$R_{H}=10^{5} cm^{-1}$$, $$h=6.6 \times 10^{-34}$$ Js,$$c= 3\times 10^{8}$$ m/s
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 69
The d- electronic configuration of an octahedral Co(II) complex having magnetic moment of 3.95 BM is:
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 70
The correct stability order of the following species/molecules is: