NTA JEE Mains 23rd Jan 2025 Shift 1
For the following questions answer them individually
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 51
Given below are two statements:
Statement I: Fructose does not contain an aldehydic group but still reduces Tollen's reagent
Statement II: In the presence of base, fructose undergoes rearrangement to give glucose. In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 52
The complex that shows Facial - Meridional isomerism is:
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 53
$$FeO_{4}^{2-}\xrightarrow{+2.0_{V}} Fe^{3+}\xrightarrow{0.8_{V}} Fe^{2+}\xrightarrow{-0.5_{V}}Fe^{0}$$ In the above diagram, the standard electrode potentials are given in volts (over the arrow). The value of $$E_{FeO_{4}^{2-}/Fe^{2+}}^{0}$$ is
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 54
The element that does not belong to the same period of the remaining elements (modern periodic table) is:
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 55
Match the LIST-I with LIST-II
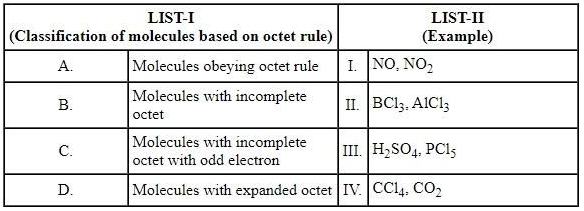
Choose the correct answer from the options given below:
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 56
What amount of bromine will be required to convert 2 g of phenol into 2,4,6-tribromophenol? (Given molar mass in $$gmol^{-1}$$ of C, H, O, Br are 12, 1, 16, 80 respectively)
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 57
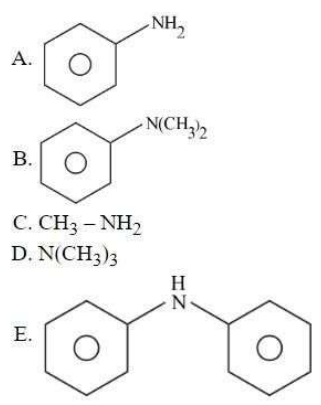
Which among the following react with Hinsberg's reagent?
Choose the correct answer from the options given below:
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 58
The correct set of ions (aqueous solution) with same colour from the following is:
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 59
Given below are two statements:
Statement I: In Lassaigne's test, the covalent organic molecules are transformed into ionic compounds.
Statement II: The sodium fusion extract of an organic compound having N and S gives prussian blue colour with $$FeSO_{4}$$ and $$Na_{4}[Fe(CN)_{6}]$$ In the light of the above statements, choose
the correct answer from the options given below.
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 60
Propane molecule on chlorination under photochemical condition gives two di-chloro products, " x " and " y ". Amongst " x " and " y ", " x " is an optically active molecule. How many tri-chloro products (consider only structural isomers) will be obtained from " x " when it is further treated with chlorine under the photochemical condition?


