NTA JEE Mains 23rd Jan 2025 Shift 1
For the following questions answer them individually
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 71
The standard enthalpy and standard entropy of decomposition of $$N_{2}O_{4}$$ to $$NO_{2}$$ are 55.0 kJ $$mol^{-1}$$ and 175.0 J/K/mol respectively. The standard free energy change for this reaction at $$25^{\circ}C$$ in J $$mol^{-1}$$ is ______ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 72
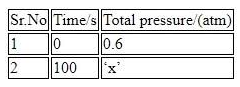
For the thermal decomposition of $$N_{2}O_{5}(g)$$ at constant volume, the following table can be formed, for the reaction mentioned below. $$2 N_{2}O_{5}(g)\rightarrow 2 N_{2}O_{4}(g)+O_{2}(g)$$
$$x= .... \times 10^{-3}$$ atm [nearest integer] Given : Rate constant for the reaction is $$4.606 \times 10^{-2} s^{-1}.$$
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 73
During " S " estimation, 160 mg of an organic compound gives 466 mg of barium sulphate. The percentage of Sulphur in the given compound is _______ %. (Given molar mass in $$gmol^{-1}$$ of Ba : 137, S : 32, O : 16)
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 74
If 1 mM solution of ethylamine produces pH=9, then the ionization constant $$(K_{b})$$ of ethylamine is $$10^{-x}$$. The value of is ______ (nearest integer). [The degree of ionization of ethylamine can be neglected with respect to unity.]
789
456
123
0.-
Clear All
NTA JEE Mains 23rd Jan 2025 Shift 1 - Question 75
Consider the following sequence of reactions to produce major product (A)
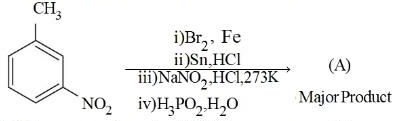
Molar mass of product (A) is $$gmol^{-1}$$. (Given molar mass in $$gmol^{-1}$$ of C : 12, H : 1, O : 16, Br : 80, N : 14, P : 31)
789
456
123
0.-
Clear All


