NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 51
Given below are two statements : Statement (I) : Nitrogen, sulphur, halogen and phosphorus present in an organic compound are detected by Lassaigne's Test. Statement (II) : The elements present in the compound are converted from covalent form into ionic form by fusing the compound with Magnesium in Lassaigne's test. In the light of the above statements, choose the correct anower from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 52
Density of 3 M NaCl solution is $$1.25 g/mL$$. The molality of the solution is :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 53
The correct order of the following complexes in terms of their crystal field stabilization energies is :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 54
Given below are two statements : Statement (I) : Corrosion is an electrochemical phenomenon in which pure metal acts as an anode and impure metal as a cathode. Statement (II) : The rate of corrosion is more in alkaline medium than in acidic medium. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 55
Consider the given figure and choose the correct option :
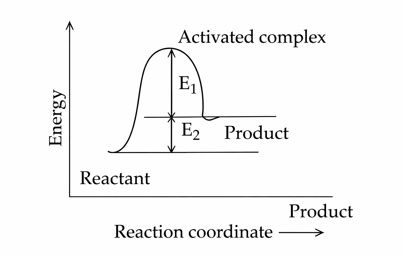
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 56
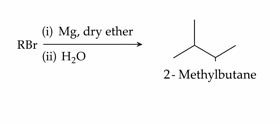
The maximum number of RBr producing 2-methylbutane by above sequence of reactions is ________ -
(Consider the structural isomers only)
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 57
The species which does not undergo disproportionation reaction is :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 58
The molar solubility(s) of zirconium phosphate with molecular formula $$(Zr^{4+})_{3}(PO_{4}^{3-})_{4}$$ is given by relation :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 59
Identify the homoleptic complex(es) that is/are low spin. $$(A)[Fe(CN)_{5}NO]^{2-}(B)[CoF_{6}]^{3-}(C)[Fe(CN)_{6}]^{4-}(D)[Co(NH_{3})_{6}]^{3+}(E)[Cr(H_{2}O)_{6}]^{2+}$$ Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 60

Match List - I with List - II.
Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 61
Identify the number of structure/s from the following which can be correlated to D-glyceraldehyde.
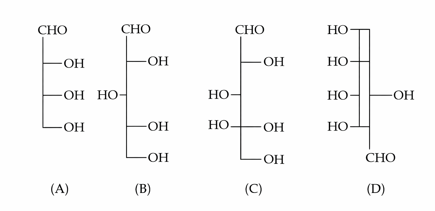
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 62
Given below are two statements : Statement (I) : A spectral line will be observed for a $$2p_{x}\rightarrow 2p_{y}$$ transition.Statement (II) :$$2p_{x}$$ and $$2p_{y}$$ are degenerate orbitals. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 63
Given below are two statements : Statement (I) : An element in the extreme left of the periodic table forms acidic oxides. Statement (II) : Acid is formed during the reaction between water and oxide of a reactive element present in the extreme right of the periodic table. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 64
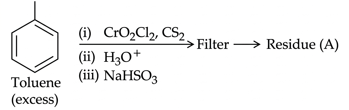
Residue (A) + HCl (dil)$$\rightarrow $$ Compound (B) Structure of residue (A) and Compound (B) formed respectively is :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 65
The alkane from below having two secondary hydrogens is :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 66
When sec-butylcyclohexane reacts with bromine in the presence of sunlight, the major product is :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 67
The most stable carbocation from the following is :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 68
Match the Compounds (List - I) with the appropriate Catalyst/Reagents (List - II) for their reduction into corresponding amines.
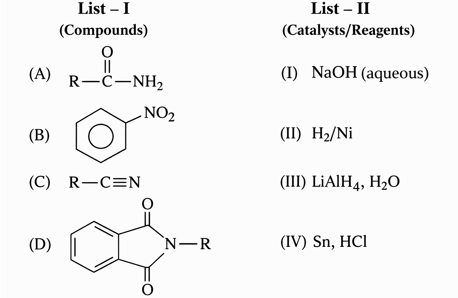
Choose the correct answer from the options given below :
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 69
Arrange the following compounds in increasing order of their dipole moment : $$HBr,H_{2}S,NF_{3}\text{ and }CHCl_{3}$$
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 70
The maximum covalency of a non-metallic group 15 element ' E ' with weakest E-E bond is:
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 71
The compound with molecular formula $$C_{6}H_{6}$$, which gives only one monobromo derivative and takes up four moles of hydrogen per mole for complete hydrogenation has ________ electrons.
789
456
123
0.-
Clear All
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 72
20 mL of 2 M NaOH solution is added to 400 mL of 0.5 M NaOH solution. The final concentration of the solution is _______ $$\times 10^{-2}M$$.(Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 73
Consider the following cases of standard enthalpy of reaction ($$\Delta H_{r}^{\circ}$$ in kJ $$mol^{-1}$$)
$$C_{2}H_{6}(g)+\frac{7}{2}O_{2}(g) \rightarrow 2CO_{2}(g)+3H_{2}O(1)\Delta H_{1}^{\circ}=-1550$$
$$C(graphite)+O_{2}(g)\rightarrow CO_{2}(g)$$ $$\Delta H_{2}^{\circ}=-393.5$$ The magnitude of $$\Delta H_{fC_{2}H_{6}(g)}^{\circ}$$ is_______
$$H_{2}(g)+\frac{1}{2}O_{2}(g)\rightarrow H_{2}O(1)$$ $$\Delta H_{3}^{\circ}=-286$$
$$kJ mol^{-1}$$ (Nearest integer).
789
456
123
0.-
Clear All
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 74
Niobium ( Nb ) and ruthenium (Ru) have "x" and "y" number of electrons in their respective 4 d orbitals The value of x+y is _______.
789
456
123
0.-
Clear All
NTA JEE Mains 22nd Jan 2025 Shift 2 - Chemistry - Question 75
The complex of $$Ni^{2+}$$ ion and dimethyl glyoxime contains ________ number of Hydrogen (H) atoms.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




