NTA JEE Mains 21st Jan 2026 Shift 1
For the following questions answer them individually
NTA JEE Mains 21st Jan 2026 Shift 1 - Question 61
Given below are two statements:
Statement I: The number of pairs among $$[SiO_{2},CO_{2}],[SnO,SnO_{2}],[PbO,PbO_{2]}$$ and $$[GeO,GeO_{2}]$$, which contain oxides that are both amphoteric is 2.
Statement ll: $$BF_{3}$$ is an electron deficient molecule, can act as a Lewis add, forms adduct with $$NH_{3}$$ and has a trigonal planar geometry.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 21st Jan 2026 Shift 1 - Question 62
Which of the following graphs between pressure 'p' versus volume 'V' represents the maximum work done?
NTA JEE Mains 21st Jan 2026 Shift 1 - Question 63
Identify A in the following reaction.
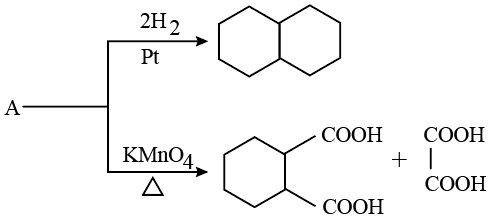
NTA JEE Mains 21st Jan 2026 Shift 1 - Question 64
A hydrocarbon 'P' $$(C_{4}H_{8})$$ on reaction with HCl gives an optically active compound 'Cl' $$(C_{4}H_{9}Cl)$$ which on reaction with one mole of ammonia gives compound 'R' $$(C_{4}H_{11}N)$$ on diazolization followed by hydrolysis gives 'S'. Identify P, Q, Rand S.
NTA JEE Mains 21st Jan 2026 Shift 1 - Question 65
Elements P and Q form two types of non-volatile, non-ionizable compounds PQ and $$PQ_{2}$$. When 1g of PQ is dissolved in 50 g of solvent ''A', $$\Delta T_{b}$$was 1.176 K while when 1 g of $$PQ_{2}$$ is dissolved in 50g of solvent 'A'.$$\Delta T_{b}$$ was 0.689 K ($$K_{b}$$ of 'A' =5K kg $$mol^{-1}$$) The molar masses of elements P and Q (in g $$mol^{-1}$$ ) respectively, are:
NTA JEE Mains 21st Jan 2026 Shift 1 - Question 66
Identify correct statements from the following :
A Propanal and propanone are functional isomers.
B. Ethoxyethane and methoxypropane are metamers.
C. But-2-ene shows optical isomerism.
D. But-1-ene and but-2-ene are functional isomers.
E. Pentane and 2, 2-dimethyl propane ai-e chain isomers.
Choose the correct answer from the options given below:
NTA JEE Mains 21st Jan 2026 Shift 1 - Question 67
An organic compound (P) on treatment with aqueous ammonia under hot condition forms compound (Q) which on heating with $$Br_{2}$$ and KOH forms compound (R) having molecular formula $$C_{6}H_{7}N$$ Names of P, Q and R respectively are.
NTA JEE Mains 21st Jan 2026 Shift 1 - Question 68
Given below are two statements:
Statement I :
The number of species among $$SF_{4},NH_4^+,[NiCl_{4}]^{2-},XeF_{4},[PtCl_{4}]^{2-},SeF_{4}$$ and $$[Ni(CN)_{4}]^{2-}$$, tha t have tetrahedral geometry is 3.
Statement II :
In the set $$[NO_{2},BeH_{2},BF_{3},AlCl_{3}]$$ all the molecules have incomplete octet around central atom.
In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 21st Jan 2026 Shift 1 - Question 69
In Carius method, 0.75 g of an organic compound gave 1.2 g of barium sulphate, find percentage of sulphur (molar mass 32 g $$mol^{-1}$$ ) Molar mass of barium sulphate is 233 g $$mol^{-1}$$
NTA JEE Mains 21st Jan 2026 Shift 1 - Question 70
For the reaction $$N_{2}O_{4}\rightleftharpoons2NO_{2}$$ , graph is plotted as shown below. Identify correct statements.
A. Standard free energy change for the reaction is $$-5.40kJmol^{-1}$$.
B. As $$\triangle G^{\ominus}$$ in graph is positive, $$N_{2}O_{4}$$ will not dissociate into $$NO_{2}$$ at all.
C. Reverse reaction will go to completion.
D. When 1 mole of $$N_{2}O_{4}$$ changes into equilibrium mixture, value of $$\triangle G^{\ominus}$$ = -0.84kJ $$mol^{-1}$$.
E. When 2 mole of $$NO_{2}$$ changes into equilibrium mixture, $$\triangle G^{\ominus}$$ for equilibrium mixture is -6.24kJ $$mol^{-1}$$.
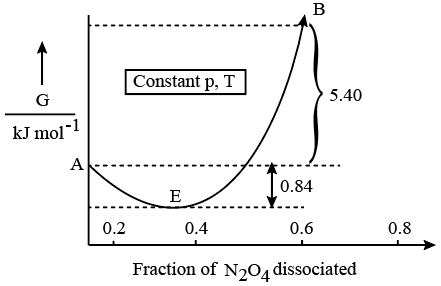
Choose the correct answer from the options given below:


