NTA JEE Mains 21st Jan 2026 Shift 1
For the following questions answer them individually
NTA JEE Mains 21st Jan 2026 Shift 1 - Question 71
Consider the following reaction sequence
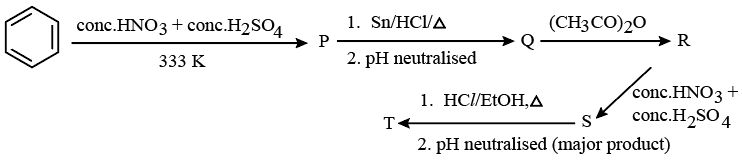
The percentage of nitrogen in product 'T' formed is ____ %. (Nearest integer)
(Given molar mass in g $$mol^{-1}$$ H : 1, C: 12, N : 14, 0: 16)
789
456
123
0.-
Clear All
NTA JEE Mains 21st Jan 2026 Shift 1 - Question 72
Pre-exponential factors of two different reactions of same order are identical. Let activation energy of first reaction exceeds the activation energy of second reaction by 20 kJ $$mol^{-1}$$. If $$k_{1}\text{ and }k_{2}$$ are the rate constants of first and second reaction respectively at 300 K, then In $$\frac{k_{2}}{k_{1}}$$ will be ___.
(nearest integer) $$[R=8.3JK^{-1}mol^{-1}]$$
789
456
123
0.-
Clear All
NTA JEE Mains 21st Jan 2026 Shift 1 - Question 73
Use the following data :
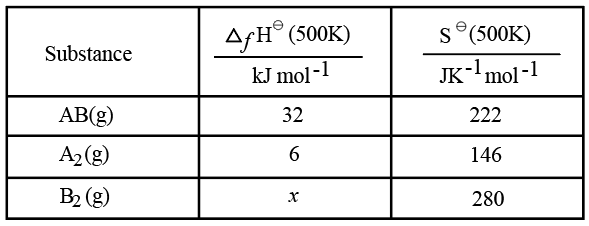
One mole each of $$A_{2}(g)$$ and $$B_{2}(g)$$ are taken in a 1 L closed flask and allowed to establish the equilibrium at 500K
$$A_{2}(g)+B_{2}(g)\rightleftharpoons2AB(g)$$
The value of x $$(in kJ mol^{-1})$$ is ____ . (Nearest integer)
(Given: log K=2.2 R= 8.3 kJ $$K^{-1} mol^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Mains 21st Jan 2026 Shift 1 - Question 74
Consider the following reactions:
$$NaCl+K_{2}Cr_{2}O_{7}+H_{2}SO_{4}\rightarrow A+KHSO_{4}+NaHSO_{4}+H_{2}O$$
$$A+NaOH\rightarrow B+NaCl+H_{2}O$$
$$B+H_{2}SO_{4}+H_{2}O_{2}\rightarrow C+Na_{2}SO_{4}+H_{2}O$$
In the product 'C, 'X' is the number of $$O_{2}^{2-}$$ units, 'Y' is the total number oxygen atoms present and 'Z' is the oxidation state of Cr·. The value of X + Y + Z is ______
789
456
123
0.-
Clear All
NTA JEE Mains 21st Jan 2026 Shift 1 - Question 75
The pH and conductance of a weak acid (HX) was found to be 5 and $$4\times10^{5}S$$. respectively. The conductance was measured under standard condition using a cell where the electrode plates having a surface area of 1 $$cm^{2}$$ were at a distance of 15 cm apart. The value of the limiting molar conductivity is ______ S $$m^{2}mol^{-1}$$ (nearest integer)
(Given : degree of dissociation of the weak acid ($$\alpha$$) < < 1)
789
456
123
0.-
Clear All


