NTA JEE Mains 2nd April Shift 2 2026
For the following questions answer them individually
NTA JEE Mains 2nd April Shift 2 2026 - Question 61
Which of the following complexes will show coordination isomerism?
A. $$[Ag(NH_3)_2][Ag(CN)_2]$$
B. $$[Co(NH_3)_6][Cr(CN)_6]$$
C. $$[Co(NH_3)_6][Co(CN)_6]$$
D. $$[Fe(NH_3)_6][Co(CN)_6]$$
E. $$[Co(NH_3)_6][Fe(CN)_6]$$
Choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 2 2026 - Question 62
Complete combustion of $$X$$ g of an organic compound gave 0.25 g of CO$$_2$$ and 0.12 g of H$$_2$$O. If the % of carbon is 25% and of hydrogen is 4.89%, then $$X = $$ _____ $$\times 10^{-3}$$ g. (Nearest integer) (Molar mass of C, H and O are 12, 1 and 16 g mol$$^{-1}$$ respectively.)
NTA JEE Mains 2nd April Shift 2 2026 - Question 63
Given below are two statements :
Statement I : In
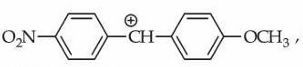
the carbocation is stabilised by +R effect of $$-OCH_3$$ group.
Statement II : In
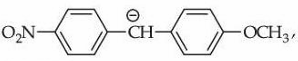
the carbanion is stabilised by $$-R$$ effect of $$-NO_2$$ group.
NTA JEE Mains 2nd April Shift 2 2026 - Question 64
The compound (X) on
(i) on heating in the presence of anhydrous AlCl$$_3$$ and HCl gas gives 2,4-dimethyl pentane
(ii) aromatization gives toluene and
(iii) cyclisation gives methyl cyclohexane
The correct name of compound (X) is :
NTA JEE Mains 2nd April Shift 2 2026 - Question 65
Correct statements regarding alkyl halides $$(R-X)$$ among the following are :
A. Alcohol being less polar solvent favours elimination with alcoholic KOH favours elimination reaction with $$R - X.$$
B. Order of reactivity towards $$S_N1$$ is $$C_6H_5-CH_2-Cl > C_6H_5-CHCl-C_6H_5$$.
C. Non substituted aryl halides exhibit properties similar to alkyl halides.
D. Vinyl chloride is example of haloalkene and allyl chloride is example of haloalkyne.
E. $$R-Cl$$ can be prepared by reacting $$R-OH$$ with $$SOCl_2$$ but $$Ar-Cl$$ cannot be prepared by reacting $$Ar-OH$$ with $$SOCl_2$$.
Choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 2 2026 - Question 66
An organic compound "x" where molar ratio of C, O and H are equal, on treatment with 50% KOH under reflux followed by acidification produced "y". The most likely structure of "y" is :
[Molar mass of 'x' is 58 g mol$$^{-1}$$]
NTA JEE Mains 2nd April Shift 2 2026 - Question 67
A molecule (X) with the following structure under mild acidic condition is hydrolysed to produce (Y) and (Z). Identify the correct statements about (Y) and (Z).
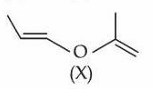
A. Both (Y) and (Z) have same molar mass.
B. (Y) and (Z) can be distinguished from each other by NaHCO$$_3$$.
C. (Y) and (Z) react with HCN with same rates.
D. (Y) and (Z) undergo addition reaction with 2,4-DNP.
Choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 2 2026 - Question 68
Identify compounds A and E in the following reaction sequence.
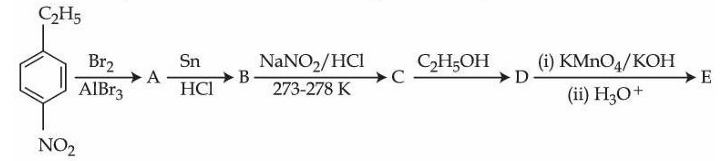
NTA JEE Mains 2nd April Shift 2 2026 - Question 69
Identify the correct pair having amino acid (A) and the hormone (B) that is iodinated derivative of the amino acid (A).
(T and Y represent one letter code for amino acids)
Amino acid (A) Hormone (B)
NTA JEE Mains 2nd April Shift 2 2026 - Question 70
Among $$Fe^{2+}$$, $$Fe^{3+}$$, $$Cr^{2+}$$ and $$Zn^{2+}$$, the ion that shows positive borax bead test and with highest ionisation enthalpy is :


