NTA JEE Mains 2nd April Shift 2 2026
For the following questions answer them individually
NTA JEE Mains 2nd April Shift 2 2026 - Question 51
The ratio of mass percentage (w/w) of C : H in a hydrocarbon is 12 : 1. It has two carbon atoms. The weight (in g) of CO$$_2$$(g) formed when 3.38 g of this hydrocarbon is completely burnt in oxygen is : (Given: Molar mass in g mol$$^{-1}$$ C : 12, H : 1, O : 16)
NTA JEE Mains 2nd April Shift 2 2026 - Question 52
The first and second ionization constants of a weak dibasic acid $$H_2A$$ are $$8.1 \times 10^{-8}$$ and $$1.0 \times 10^{-13}$$ respectively. 0.1 mol of $$H_2A$$ was dissolved in 1 L of 0.1 M HCl solution. The concentration of $$HA^-$$ in the resultant solution is :
NTA JEE Mains 2nd April Shift 2 2026 - Question 53
$$SF_4$$ is isostructural with :
A. $$BrF_4^\ominus$$
B. $$CH_4$$
C. $$IF_4^\oplus$$
D. $$XeF_4$$
E. $$XeO_2F_2$$
Choose the correct answer from the options given below :
NTA JEE Mains 2nd April Shift 2 2026 - Question 54
Gas 'A' undergoes change from state 'X' to state 'Y'. In this process, the heat absorbed and work done by the gas is 10 J and 18 J respectively. Now gas is brought back to state 'X' by another process during which 6 J of heat is evolved. In the reverse process of 'Y' to 'X' :
NTA JEE Mains 2nd April Shift 2 2026 - Question 55
Solution A is prepared by dissolving 1 g of a protein (molar mass = 50000 g mol$$^{-1}$$) in 0.5 L of water at 300 K. Its osmotic pressure is $$x$$ bar. Solution B is made by dissolving 2 g of same protein in 1 L of water at 300 K. Osmotic pressure of solution B is $$y$$ bar. Entire solution of A is mixed with entire solution of B at same temperature. The osmotic pressure of resultant solution is $$z$$ bar. $$x$$, $$y$$ and $$z$$ respectively are :
($$R = 0.083$$ L bar mol$$^{-1}$$ K$$^{-1}$$)
NTA JEE Mains 2nd April Shift 2 2026 - Question 56
At 25°C, 20.0 mL of 0.2 M weak monoprotic acid HX is titrated against 0.2 M NaOH. The pH of the solution (a) at the start of the titration (when NaOH has not been added) and (b) when 10 mL of NaOH is added respectively, are :
Given: $$K_a = 5 \times 10^{-4}$$,
$$pK_a = 3.3$$,
$$\alpha \ll 1$$
NTA JEE Mains 2nd April Shift 2 2026 - Question 57
Consider the reaction $$aX \to bY$$, for which the rate constant at 30°C is $$1 \times 10^{-3}$$ mol$$^{-1}$$ L s$$^{-1}$$. Which of the following statements are true?
A. When concentration of 'X' is increased to four times, the rate of reaction becomes 16 times.
B. The reaction is a second order reaction.
C. The half-life period is independent of the concentration of X.
D. Decomposition of $$N_2O_5$$ is an example of the above reaction.
E.
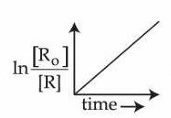
is valid for the above reaction.
Choose the correct answer from the option given below:
NTA JEE Mains 2nd April Shift 2 2026 - Question 58
The correct set that contains all kinds (basic, acidic, amphoteric and neutral) of oxides is :
NTA JEE Mains 2nd April Shift 2 2026 - Question 59
Given below are two statements :
Statement I : The second ionization enthalpy of B, Al and Ga is in the order of $$B > Al > Ga$$.
Statement II : The correct order in terms of first ionization enthalpy is $$Si < Ge < Pb < Sn$$.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 2nd April Shift 2 2026 - Question 60
Given below are two statements :
Statement I : Among Zn, Mn, Sc and Cu, the energy required to remove the third valence electron is highest for Zn and lowest for Sc.
Statement II : The correct order of the following complexes in terms of CFSE is $$[Co(H_2O)_6]^{2+} < [Co(H_2O)_6]^{3+} < [Co(en)_3]^{3+}$$.
In the light of the above statements, choose the correct answer from the options given below:


