NTA JEE Mains 2nd April Shift 2 2026
For the following questions answer them individually
NTA JEE Mains 2nd April Shift 2 2026 - Question 71
The surface of sodium metal is irradiated with radiation of wavelength $$x$$ nm. The kinetic energy of ejected electrons is $$2.8 \times 10^{-20}$$ J. The work function of sodium is 2.3 eV. The value of $$x$$ is _____ $$\times 10^2$$ nm. (Nearest integer) (Given: $$h = 6.6 \times 10^{-34}$$ J s; $$1$$ eV $$= 1.6 \times 10^{-19}$$ J; $$c = 3.0 \times 10^8$$ m s$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Mains 2nd April Shift 2 2026 - Question 72
Consider the following gas phase reaction being carried out in a closed vessel at 25°C.
$$2A(g) \to 4B(g) + C(g)$$

The pressure of $$C(g)$$ at 30 minutes time interval would be _____ mm Hg. (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 2nd April Shift 2 2026 - Question 73
Consider the following two half-cell reactions along with the standard reduction potential given :
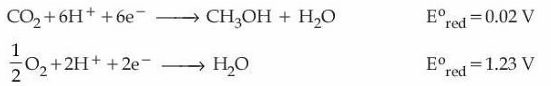
A fuel cell was set up using the above two reactions such that the cell operates under the standard condition of 1 bar pressure and 298 K temperature. The fuel cell works with 80% efficiency. If the work derived from the cell using 1 mol of CH$$_3$$OH is used to compress an ideal gas isothermally against a constant pressure of 1 kPa, then the change in the volume of the gas, $$\Delta V = $$ _____ m$$^3$$. (nearest integer)
Given: $$F = 96500$$ C mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Mains 2nd April Shift 2 2026 - Question 74
Number of paramagnetic ions among the following d- and f-block metal ions is _________.
$$Mn^{2+}$$, $$Cu^{2+}$$, $$Zn^{2+}$$, $$Yb^{2+}$$, $$Sc^{3+}$$, $$La^{3+}$$, $$Gd^{3+}$$, $$Lu^{3+}$$, $$Ti^{4+}$$, $$Ce^{4+}$$
(Atomic number of Mn = 25, Cu = 29, Zn = 30, Yb = 70, Sc = 21, La = 57, Gd = 64, Lu = 71, Ti = 22, Ce = 58)
789
456
123
0.-
Clear All
NTA JEE Mains 2nd April Shift 2 2026 - Question 75
Consider the following reactions sequence.
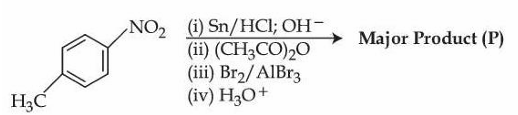
When the product (P) is subjected to Carius analysis using AgNO$$_3$$, 1.0 g of the product (P) will produce _____ g of the precipitate of AgBr. (Nearest Integer)
(Given: molar mass in g mol$$^{-1}$$ C: 12, H: 1, O: 16, N: 14, Br: 80, Ag: 108)
789
456
123
0.-
Clear All


