NTA JEE Mains 1st Feb 2023 Shift 1
For the following questions answer them individually
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 41
A solution of $$FeCl_3$$ when treated with $$K_4[Fe(CN)_6]$$ gives a prussian blue precipitate due to the formation of
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 42
Highest oxidation state of Mn is exhibited in $$Mn_2O_7$$. The correct statements about $$Mn_2O_7$$ are
(A) Mn is tetrahedrally surrounded by oxygen atoms
(B) Mn is octahedrally surrounded by oxygen atoms
(C) Contains Mn - O - Mn bridge
(D) Contains Mn - Mn bond.
Choose the correct answer from the options given below
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 43
Which of the following complex will show largest splitting of d-orbitals?
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 44
Which of the following are the example of double salt?
(A) $$FeSO_4 \cdot (NH_4)_2SO_4 \cdot 6H_2O$$
(B) $$CuSO_4 \cdot 4NH_3 \cdot H_2O$$
(C) $$K_2SO_4 \cdot Al_2(SO_4)_3 \cdot 24H_2O$$
(D) $$Fe(CN)_2 \cdot 4KCN$$
Choose the correct answer.
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 45
Identify the incorrect option from the following:
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 46
Decreasing order of dehydration of the following alcohols is
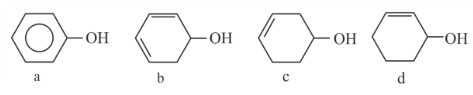
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 47
In the following reaction, 'A' is:
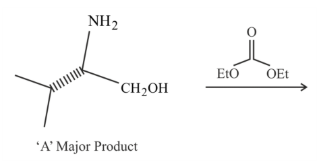
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 48
Match List I with List II

Choose the correct answer from the options given below:
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 49
The correct representation in six membered pyranose form for the following sugar [X] is:
Sugar [X] has the Fischer projection: CHO, HO-H, HO-H, H-OH, H-OH, $$H_2COH$$
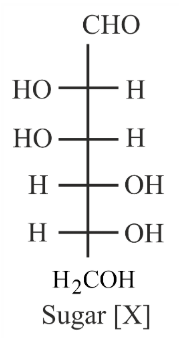
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 50
Match List I and List II

Choose the correct answer from the options given below:


