NTA JEE Mains 1st Feb 2023 Shift 1
For the following questions answer them individually
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 31
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R
Assertion A: Hydrogen is an environment friendly fuel.
Reason R: Atomic number of hydrogen is 1 and it is a very light element.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 32
Match List I with List II

Choose the correct answer form the options given below:
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 33
Choose the correct statement(s):
A. Beryllium oxide is purely acidic in nature.
B. Beryllium carbonate is kept in the atmosphere of $$CO_2$$.
C. Beryllium sulphate is readily soluble in water.
D. Beryllium shows anomalous behavior.
Choose the correct answer from the options given below:
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 34
Resonance in carbonate ion $$CO_3^{2-}$$ i
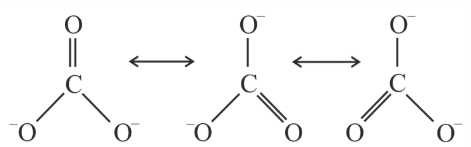
shown with three resonance structures. Which of the following is true?
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 35
But-2-yne is reacted separately with one mole of Hydrogen as shown below:
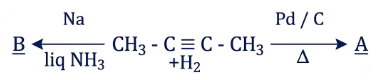
Identify the incorrect statements from the options given below:
A. A is more soluble than B.
B. The boiling point & melting point of A are higher and lower than B respectively.
C. A is more polar than B because dipole moment of A is zero.
D. $$Br_2$$ adds easily to B than A.
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 36
How can photochemical smog be controlled?
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 37
Which of the following represents the lattice structure of $$A_{0.95}O$$ containing $$A^{2+}, A^{3+}$$ and $$O^{2-}$$ ions
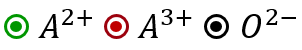
(A)
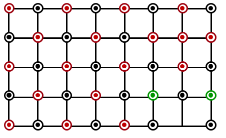
(B)
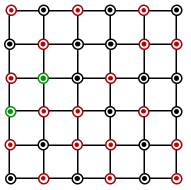
(c)
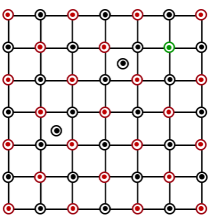
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 38
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Amongst He, Ne, Ar and Kr; 1 g of activated charcoal adsorbs more of Kr.
Reason R: The critical volume $$V_c$$ $$(cm^3 mol^{-1})$$ and critical pressure $$P_c$$ (atm) is highest for Krypton but the compressibility factor at critical point $$Z_c$$ is lowest for Krypton.
In the light of the above statements, choose the correct answer from the options given below.
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 39
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R
Assertion A: In an Ellingham diagram, the oxidation of carbon to carbon monoxide shows a negative slope with respect to temperature.
Reason R: CO tends to get decomposed at higher temperature.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 40
Given below are two statements:
Statement I: Chlorine can easily combine with oxygen to form oxides: and the product has a tendency to explode.
Statement II: Chemical reactivity of an element can be determined by its reaction with oxygen and halogens.
In the light of the above statements, choose the correct answer from the options given below


