NTA JEE Mains 1st Feb 2023 Shift 1
For the following questions answer them individually
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 51
The density of 3M solution of NaCl is $$1.0 \text{ g mL}^{-1}$$. Molality of the solution is ______ $$\times 10^{-2}$$ m (Nearest integer).
Given: Molar mass of Na and Cl is 23 and 35.5 $$\text{g mol}^{-1}$$ respectively.
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 52
Electrons in a cathode ray tube have been emitted with a velocity of $$1000 \text{ ms}^{-1}$$. The number of following statements which is/are true about the emitted radiation is
Given: $$h = 6 \times 10^{-34}$$ Js, $$m_e = 9 \times 10^{-31}$$ kg
(A) The deBroglie wavelength of the electron emitted is 666.67 nm
(B) The characteristic of electrons emitted depend upon the material of the electrodes of the cathode ray tube.
(C) The cathode rays start from cathode and move towards anode.
(D) The nature of the emitted electrons depends on the nature of the gas present in cathode ray tube.
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 53
At 25°C, the enthalpy of the following processes are given:
$$H_2(g) + O_2(g) \rightarrow 2OH(g) \quad \Delta H^o = 78 \text{ kJ mol}^{-1}$$
$$H_2(g) + \frac{1}{2}O_2(g) \rightarrow H_2O(g) \quad \Delta H^o = -242 \text{ kJ mol}^{-1}$$
$$H_2(g) \rightarrow 2H(g) \quad \Delta H^o = 436 \text{ kJ mol}^{-1}$$
$$\frac{1}{2}O_2(g) \rightarrow O(g) \quad \Delta H^o = 249 \text{ kJ mol}^{-1}$$
What would be the value of X for the following reaction? (Nearest integer)
$$H_2O(g) \rightarrow H(g) + OH(g) \quad \Delta H^o = X \text{ kJ mol}^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 54
(i) $$X(g) \rightleftharpoons Y(g) + Z(g) \quad K_{p1} = 3$$
(ii) $$A(g) \rightleftharpoons 2B(g) \quad K_{p2} = 1$$
If the degree of dissociation and initial concentration of both the reactants $$X(g)$$ and $$A(g)$$ are equal, then the ratio of the total pressure at equilibrium $$\frac{p_1}{p_2}$$ is equal to x : 1. The value of x is (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 55
The total number of chiral compound/s from the following is
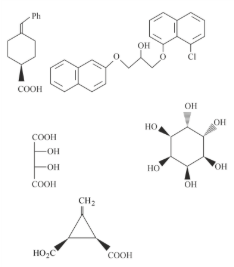
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 56
25 mL of an aqueous solution of KCl was found to require 20 mL of 1M $$AgNO_3$$ solution when titrated using $$K_2CrO_4$$ as an indicator. What is the depression in freezing point of KCl solution of the given concentration? (Nearest integer).
(Given: $$K_f = 2.0 \text{ K kg mol}^{-1}$$)
Assume 1) 100% ionization and 2) density of the aqueous solution as $$1 \text{ g mL}^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 57
At what pH, given half cell $$MnO_4^-(0.1 M) | Mn^{2+}(0.001 M)$$ will have electrode potential of 1.282 V? (Nearest Integer)
Given $$E^o_{MnO_4^-/Mn^{2+}} = 1.54$$ V, $$\frac{2.303RT}{F} = 0.059$$ V
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 58
A and B are two substances undergoing radioactive decay in a container. The half life of A is 15 min and that of B is 5 min. If the initial concentration of B is 4 times that of A and they both start decaying at the same time, how much time will it take for the concentration of both of them to be same? ______ min.
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 59
Sum of oxidation states of bromine in bromic acid and perbromic acid is
789
456
123
0.-
Clear All
NTA JEE Mains 1st Feb 2023 Shift 1 - Question 60
Number of isomeric compounds with molecular formula $$C_9H_{10}O$$ which
(i) do not dissolve in NaOH
(ii) do not dissolve in HCl.
(iii) do not give orange precipitate with 2, 4 - DNP
(iv) on hydrogenation give identical compound with molecular formula $$C_9H_{12}O$$ is
789
456
123
0.-
Clear All


