NTA JEE Mains 05th April 2024 Shift 2
For the following questions answer them individually
NTA JEE Mains 05th April 2024 Shift 2 - Question 41
The quantity of silver deposited when one coulomb charge is passed through $$AgNO_3$$ solution :
NTA JEE Mains 05th April 2024 Shift 2 - Question 42
For the electrochemical cell, if $$E^0_{(M^{2+}/M)} = 0.46$$ V and $$E^0_{(X/X^{2-})} = 0.34$$ V. Which of the following is correct?
NTA JEE Mains 05th April 2024 Shift 2 - Question 43
The number of ions from the following that have the ability to liberate hydrogen from a dilute acid is ______. $$Ti^{2+}, Cr^{2+}$$ and $$V^{2+}$$
NTA JEE Mains 05th April 2024 Shift 2 - Question 44
While preparing crystals of Mohr's salt, dil $$H_2SO_4$$ is added to a mixture of ferrous sulphate and ammonium sulphate, before dissolving this mixture in water, dil $$H_2SO_4$$ is added here to :
NTA JEE Mains 05th April 2024 Shift 2 - Question 45
The number of complexes from the following with no electrons in the $$t_2$$ orbital is $$TiCl_4$$, $$[MnO_4]^-$$, $$[FeO_4]^{2-}$$, $$[FeCl_4]^-$$, $$[CoCl_4]^{2-}$$
NTA JEE Mains 05th April 2024 Shift 2 - Question 46
The metal atom present in the complex MABXL (where A, B, X and L are unidentate ligands and M is metal) involves $$sp^3$$ hybridization. The number of geometrical isomers exhibited by the complex is:
NTA JEE Mains 05th April 2024 Shift 2 - Question 47
Identify the major product in the following reaction.
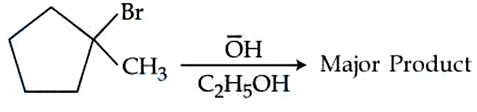
NTA JEE Mains 05th April 2024 Shift 2 - Question 48
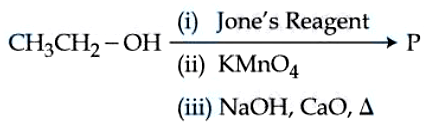
Consider the above reaction sequence and identify the major product $$P$$.
NTA JEE Mains 05th April 2024 Shift 2 - Question 49
Which one of the following reactions is NOT possible?
NTA JEE Mains 05th April 2024 Shift 2 - Question 50
Coagulation of egg, on heating is because of :


