NTA JEE Mains 05th April 2024 Shift 2
For the following questions answer them individually
NTA JEE Mains 05th April 2024 Shift 2 - Question 51
In an atom, total number of electrons having quantum numbers $$n = 4$$, $$|m_l| = 1$$ and $$m_s = -\frac{1}{2}$$ is ______
789
456
123
0.-
Clear All
NTA JEE Mains 05th April 2024 Shift 2 - Question 52
Number of compounds from the following with zero dipole moment is ______ $$HF$$, $$H_2$$, $$H_2S$$, $$CO_2$$, $$NH_3$$, $$BF_3$$, $$CH_4$$, $$CHCl_3$$, $$SiF_4$$, $$H_2O$$, $$BeF_2$$
789
456
123
0.-
Clear All
NTA JEE Mains 05th April 2024 Shift 2 - Question 53
Combustion of 1 mole of benzene is expressed at $$C_6H_6(l) + \frac{15}{2}O_2(g) \rightarrow 6CO_2(g) + 3H_2O(l)$$. The standard enthalpy of combustion of 2 mol of benzene is $$-'x'$$ kJ. $$x =$$ ______ Given: 1. Standard enthalpy of formation of 1 mol of $$C_6H_6(l)$$, for the reaction $$6C \text{(graphite)} + 3H_2(g) \rightarrow C_6H_6(l)$$ is $$48.5 \text{ kJ mol}^{-1}$$. 2. Standard enthalpy of formation of 1 mol of $$CO_2(g)$$, for the reaction $$C \text{(graphite)} + O_2(g) \rightarrow CO_2(g)$$ is $$-393.5 \text{ kJ mol}^{-1}$$. 3. Standard enthalpy of formation of 1 mol of $$H_2O(l)$$, for the reaction $$H_2(g) + \frac{1}{2}O_2(g) \rightarrow H_2O(l)$$ is $$-286 \text{ kJ mol}^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Mains 05th April 2024 Shift 2 - Question 54
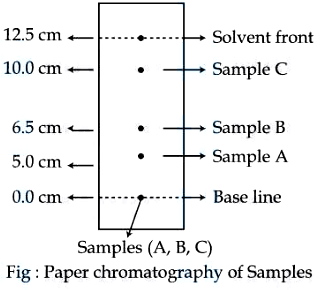
Using the given figure, the ratio of $$R_f$$ values of sample A and sample C is $$x \times 10^{-2}$$. Value of $$x$$ is _____
789
456
123
0.-
Clear All
NTA JEE Mains 05th April 2024 Shift 2 - Question 55
Considering acetic acid dissociates in water, its dissociation constant is $$6.25 \times 10^{-5}$$. If $$5$$ mL of acetic acid is dissolved in 1 litre water, the solution will freeze at $$-x \times 10^{-2} °C$$, provided pure water freezes at $$0°C$$. $$x =$$ ______ . (Nearest integer) Given : $$(K_f)_{water} = 1.86 \text{ K kg mol}^{-1}$$, density of acetic acid is $$1.2 \text{ g mL}^{-1}$$, molar mass of water $$= 18 \text{ g mol}^{-1}$$, molar mass of acetic acid $$= 60 \text{ g mol}^{-1}$$, density of water $$= 1 \text{ g cm}^{-3}$$. Acetic acid dissociates as $$CH_3COOH \rightleftharpoons CH_3COO^- + H^+$$
789
456
123
0.-
Clear All
NTA JEE Mains 05th April 2024 Shift 2 - Question 56
Consider the following single step reaction in gas phase at constant temperature. $$2A_{(g)} + B_{(g)} \rightarrow C_{(g)}$$. The initial rate of the reaction is recorded as $$r_1$$ when the reaction starts with $$1.5$$ atm pressure of A and $$0.7$$ atm pressure of B. After some time, the rate $$r_2$$ is recorded when the pressure of C becomes $$0.5$$ atm. The ratio $$r_1 : r_2$$ is ______ $$\times 10^{-1}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 05th April 2024 Shift 2 - Question 57
The fusion of chromite ore with sodium carbonate in the presence of air leads to the formation of products A and B along with the evolution of $$CO_2$$. The sum of spin-only magnetic moment values of A and B is ______ B.M. (Nearest integer) [Given atomic number : C : 6, Na : 11, O : 8, Fe : 26, Cr : 24]
789
456
123
0.-
Clear All
NTA JEE Mains 05th April 2024 Shift 2 - Question 58
In the Claisen-Schmidt reaction to prepare $$351$$ g of dibenzalacetone using $$87$$ g of acetone, the amount of benzaldehyde required is ______ g. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Mains 05th April 2024 Shift 2 - Question 59
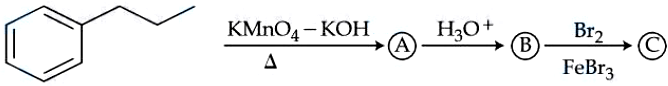
The product (C) in the following sequence of reactions has ______ $$\pi$$ bonds.
789
456
123
0.-
Clear All
NTA JEE Mains 05th April 2024 Shift 2 - Question 60
$$X$$ g of ethanamine was subjected to reaction with $$NaNO_2/HCl$$ followed by hydrolysis to liberate $$N_2$$ and HCl. The HCl generated was completely neutralised by 0.2 moles of NaOH. $$X$$ is ______ g.
789
456
123
0.-
Clear All


