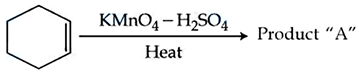NTA JEE Mains 05th April 2024 Shift 2
For the following questions answer them individually
NTA JEE Mains 05th April 2024 Shift 2 - Question 31
The number of moles of methane required to produce $$11$$ g $$CO_2(g)$$ after complete combustion is : (Given molar mass of methane in $$g mol^{-1} : 16$$)
NTA JEE Mains 05th April 2024 Shift 2 - Question 32
Given below are two statements : Statement I : The metallic radius of Na is $$1.86$$ Å and the ionic radius of $$Na^+$$ is lesser than $$1.86$$ Å. Statement II : Ions are always smaller in size than the corresponding elements. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 05th April 2024 Shift 2 - Question 33
Match List I with List II :

Choose the correct answer from the options given below :
NTA JEE Mains 05th April 2024 Shift 2 - Question 34
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R). Assertion (A) : $$NH_3$$ and $$NF_3$$ molecule have pyramidal shape with a lone pair of electrons on nitrogen atom. The resultant dipole moment of $$NH_3$$ is greater than that of $$NF_3$$. Reason (R) : In $$NH_3$$, the orbital dipole due to lone pair is in the same direction as the resultant dipole moment of the $$N-H$$ bonds. $$F$$ is the most electronegative element. In the light of the above statements, choose the correct answer from the options given below :
NTA JEE Mains 05th April 2024 Shift 2 - Question 35
Given below are two statements : Statement I : On passing $$HCl_{(g)}$$ through a saturated solution of $$BaCl_2$$, at room temperature white turbidity appears. Statement II : When HCl gas is passed through a saturated solution of NaCl, sodium chloride is precipitated due to common ion effect. In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Mains 05th April 2024 Shift 2 - Question 36
The correct statements from the following are : (A) The decreasing order of atomic radii of group 13 elements is $$Tl > In > Ga > Al > B$$. (B) Down the group 13 electronegativity decreases from top to bottom. (C) Al dissolves in dil. HCl and liberates $$H_2$$ but conc. $$HNO_3$$ renders Al passive by forming a protective oxide layer on the surface. (D) All elements of group 13 exhibits highly stable +1 oxidation state. (E) Hybridisation of Al in $$[Al(H_2O)_6]^{3+}$$ ion is $$sp^3d^2$$. Choose the correct answer from the options given below :
NTA JEE Mains 05th April 2024 Shift 2 - Question 37
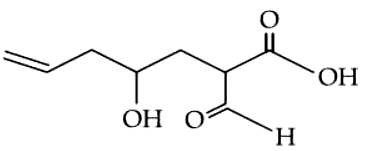
The correct nomenclature for the following compound is
NTA JEE Mains 05th April 2024 Shift 2 - Question 38
Match List I with List II :

Choose the correct answer from the options given below :
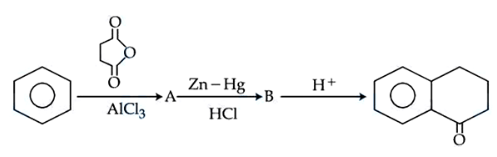
NTA JEE Mains 05th April 2024 Shift 2 - Question 39
Identify $$A$$ and $$B$$ in the given chemical reaction sequence
NTA JEE Mains 05th April 2024 Shift 2 - Question 40
Consider the given chemical reaction :