NTA JEE Mains 04th April 2024 Shift 2
For the following questions answer them individually
NTA JEE Mains 04th April 2024 Shift 2 - Question 41
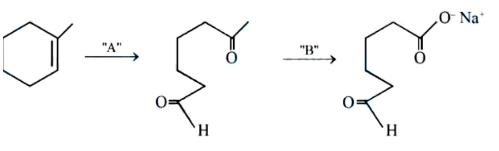
In the above chemical reaction sequence "A" and "B" respectively are:
NTA JEE Mains 04th April 2024 Shift 2 - Question 42
For a strong electrolyte, a plot of molar conductivity against (concentration)$$^{1/2}$$ is a straight line, with a negative slope, the correct unit for the slope is:
NTA JEE Mains 04th April 2024 Shift 2 - Question 43
Fuel cell, using hydrogen and oxygen as fuels: A. has been used in spaceship, B. has an efficiency of 40% to produce electricity, C. uses aluminum as catalysts, D. is eco-friendly, E. is actually a type of Galvanic cell only. Choose the correct answer from the options given below:
NTA JEE Mains 04th April 2024 Shift 2 - Question 44
When $$MnO_2$$ and $$H_2SO_4$$ is added to a salt (A), the greenish yellow gas liberated as salt (A) is:
NTA JEE Mains 04th April 2024 Shift 2 - Question 45
A first row transition metal in its +2 oxidation state has a spin-only magnetic moment value of $$3.86$$ BM. The atomic number of the metal is:
NTA JEE Mains 04th April 2024 Shift 2 - Question 46
If an iron (III) complex with the formula $$[Fe(NH_3)_x(CN)_y]^-$$ has no electron in its $$e_g$$ orbital, then the value of $$x + y$$ is:
NTA JEE Mains 04th April 2024 Shift 2 - Question 47
The number of unpaired d-electrons in $$[Co(H_2O)_6]^{3+}$$ is:
NTA JEE Mains 04th April 2024 Shift 2 - Question 48
$$CH_3-CH_2-CH_2-Br + NaOH \xrightarrow{C_2H_5OH}$$ Product 'A'.
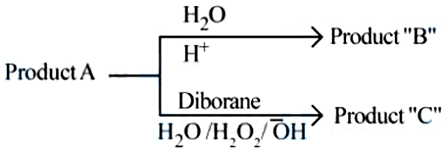
Consider the above reactions, identify product $$B$$ and product $$C$$.
NTA JEE Mains 04th April 2024 Shift 2 - Question 49
Find out the major product formed from the following reaction. [Me: $$-CH_3$$]
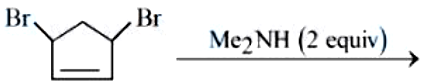
NTA JEE Mains 04th April 2024 Shift 2 - Question 50
Match List I with List II:
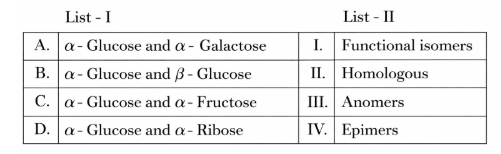
Choose the correct answer from the options given below:


