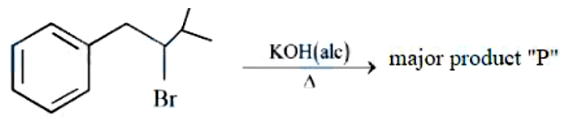NTA JEE Mains 04th April 2024 Shift 2
For the following questions answer them individually
NTA JEE Mains 04th April 2024 Shift 2 - Question 31
Choose the Incorrect Statement about Dalton's Atomic Theory
NTA JEE Mains 04th April 2024 Shift 2 - Question 32
The correct order of the first ionization enthalpy is
NTA JEE Mains 04th April 2024 Shift 2 - Question 33
Given below are two statements: Statement I: The correct order of first ionization enthalpy values of Li, Na, F and Cl is $$Na < Li < Cl < F$$. Statement II: The correct order of negative electron gain enthalpy values of Li, Na, F and Cl is $$Na < Li < F < Cl$$. In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Mains 04th April 2024 Shift 2 - Question 34
The correct statement/s about Hydrogen bonding is/are: A. Hydrogen bonding exists when H is covalently bonded to the highly electronegative atom. B. Intermolecular H bonding is present in o-nitrophenol. C. Intramolecular H bonding is present in HF. D. The magnitude of H bonding depends on the physical state of the compound. E. H-bonding has powerful effect on the structure and properties of compounds. Choose the correct answer from the options given below:
NTA JEE Mains 04th April 2024 Shift 2 - Question 35
The number of species from the following that have pyramidal geometry around the central atom is _______. $$S_2O_3^{2-}$$, $$SO_4^{2-}$$, $$SO_3^{2-}$$, $$S_2O_7^{2-}$$
NTA JEE Mains 04th April 2024 Shift 2 - Question 36
The equilibrium constant for the reaction $$SO_3(g) \rightleftharpoons SO_2(g) + \frac{1}{2}O_2(g)$$ is $$K_c = 4.9 \times 10^{-2}$$. The value of $$K_c$$ for the reaction given below is $$2SO_2(g) + O_2(g) \rightleftharpoons 2SO_3(g)$$:
NTA JEE Mains 04th April 2024 Shift 2 - Question 37
Correct order of stability of carbanion is
NTA JEE Mains 04th April 2024 Shift 2 - Question 38
Common name of Benzene-1,2-diol is:
NTA JEE Mains 04th April 2024 Shift 2 - Question 39
The adsorbent used in adsorption chromatography is/are: A. silica gel, B. alumina, C. quick lime, D. magnesia. Choose the most appropriate answer from the options given below:
NTA JEE Mains 04th April 2024 Shift 2 - Question 40