NTA JEE Mains 1st Feb 2024 Shift 1
For the following questions answer them individually
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 1
The radius $$r$$, length $$l$$ and resistance $$R$$ of a metal wire was measured in the laboratory as $$r = 0.35 \pm 0.05$$ cm, $$R = 100 \pm 10$$ ohm, $$l = 15 \pm 0.2$$ cm. The percentage error in resistivity of the material of the wire is :
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 2
The dimensional formula of angular impulse is :
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 3
A particle moving in a circle of radius $$R$$ with uniform speed takes time $$T$$ to complete one revolution. If this particle is projected with the same speed at an angle $$\theta$$ to the horizontal, the maximum height attained by it is equal to $$4R$$. The angle of projection $$\theta$$ is then given by :
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 4
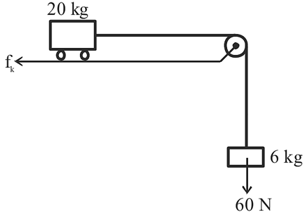
Consider a block and trolley system as shown in figure. If the coefficient of kinetic friction between the trolley and the surface is $$0.04$$, the acceleration of the system in m s$$^{-2}$$ is: (Consider that the string is massless and unstretchable and the pulley is also massless and frictionless):
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 5
A simple pendulum of length $$1$$ m has a wooden bob of mass $$1$$ kg. It is struck by a bullet of mass $$10^{-2}$$ kg moving with a speed of $$2 \times 10^{2}$$ m s$$^{-1}$$. The bullet gets embedded into the bob. The height to which the bob rises before swinging back is. (use $$g = 10$$ m s$$^{-2}$$)
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 6
A ball of mass $$0.5$$ kg is attached to a string of length $$50$$ cm. The ball is rotated on a horizontal circular path about its vertical axis. The maximum tension that the string can bear is $$400$$ N. The maximum possible value of angular velocity of the ball in rad s$$^{-1}$$ is :
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 7
If $$R$$ is the radius of the earth and the acceleration due to gravity on the surface of earth is $$g = \pi^2$$ m s$$^{-2}$$, then the length of the second's pendulum at a height $$h = 2R$$ from the surface of earth will be:
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 8
With rise in temperature, the Young's modulus of elasticity
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 9
The pressure and volume of an ideal gas are related as $$PV^{3/2} = K$$ (Constant). The work done when the gas is taken from state $$A(P_1, V_1, T_1)$$ to state $$B(P_2, V_2, T_2)$$ is :
NTA JEE Mains 1st Feb 2024 Shift 1 - Question 10
Two moles of a monoatomic gas is mixed with six moles of a diatomic gas. The molar specific heat of the mixture at constant volume is :


