NTA JEE Main 9th January 2020 Shift 2
For the following questions answer them individually
NTA JEE Main 9th January 2020 Shift 2 - Question 21
Starting at temperature $$300K$$, one mole of an ideal diatomic gas $$(\gamma = 1.4)$$ is first compressed adiabatically from volume $$V_1$$ to $$V_2 = \frac{V_1}{16}$$. It is then allowed to expand isobarically to volume $$2V_2$$. If all the processes are the quasi-static then the final temperature of the gas (in $$°K$$) is (to the nearest integer) ___________.
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 2 - Question 22
An electric field $$\vec{E} = 4x\hat{i} - (y^2 + 1)\hat{j}$$ N/C passes through the box shown in figure. The flux of the electric field through surfaces ABCD and BCGF are marked as $$\phi_I$$ and $$\phi_{II}$$ respectively. The difference between $$(\phi_I - \phi_{II})$$ is (in Nm$$^2$$/C) ___________.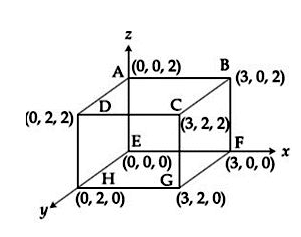
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 2 - Question 23
In a meter bridge experiment $$S$$ is a standard resistance. $$R$$ is a resistance wire. It is found that balancing length is $$l = 25$$ cm. If $$R$$ is replaced by a wire of half length and half diameter that of $$R$$ of same material, then the balancing distance $$l'$$ (in cm) will now be ___________.
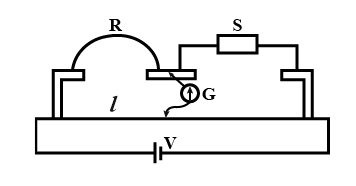
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 2 - Question 24
In a Young's double slit experiment 15 fringes are observed on a small portion of the screen when light of wavelength 500 nm is used. Ten fringes are observed on the same section of the screen when another light source of wavelength $$\lambda$$ is used. Then the value of $$\lambda$$ is (in nm) ___________.
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 2 - Question 25
In the circuit shown below, is working as a 8 V dc regulated voltage source. When 12 V is used as an input, the power dissipated (in mW) in each diode is (Considering both zener diodes are identical) ___________.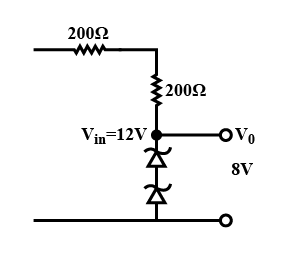
789
456
123
0.-
Clear All
NTA JEE Main 9th January 2020 Shift 2 - Question 26
The first and second ionisation enthalpies of a metal are 496 and 4560 kJ mol$$^{-1}$$, respectively. How many moles of HCl and H$$_2$$SO$$_4$$, respectively, will be needed to react completely with 1 mole of the metal hydroxide?
NTA JEE Main 9th January 2020 Shift 2 - Question 27
A mixture of gases $$O_2$$, $$H_2$$ and CO are taken in a closed vessel containing charcoal. The graph that represents the correct behaviour of pressure with time is:
NTA JEE Main 9th January 2020 Shift 2 - Question 28
The true statement amongst the following is:
NTA JEE Main 9th January 2020 Shift 2 - Question 29
In the figure shown below reactant A (represented by square) is in equilibrium with product B (represented by circle). The equilibrium constant is (approx):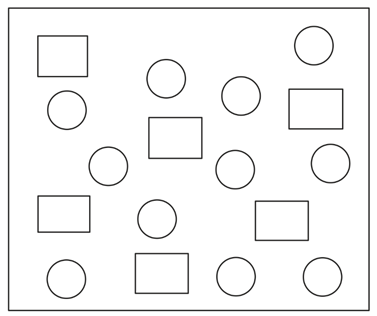
NTA JEE Main 9th January 2020 Shift 2 - Question 30
The solubility product of $$Cr(OH)_3$$ at 298K is $$6.0 \times 10^{-31}$$. The concentration of hydroxide ions in a saturated solution of $$Cr(OH)_3$$ will be:


