NTA JEE Main 6th September 2020 Shift 2
For the following questions answer them individually
NTA JEE Main 6th September 2020 Shift 2 - Question 31
Match the following:
Test/Method Reagent
(i) Lucas Test (a) $$\text{C}_6\text{H}_5\text{SO}_2\text{Cl}$$ / aq. KOH
(ii) Dumas method (b) $$\text{HNO}_3$$ / $$\text{AgNO}_3$$
(iii) Kjeldahl's method (c) CuO / $$\text{CO}_3$$
(iv) Hinsberg Test (d) Conc. HCl and $$\text{ZnCl}_2$$
(e) $$\text{H}_2\text{SO}_4$$
NTA JEE Main 6th September 2020 Shift 2 - Question 32
The IUPAC name of the following compound is: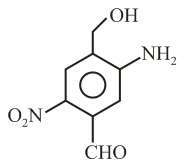
NTA JEE Main 6th September 2020 Shift 2 - Question 33
A crystal is made up of metal ions 'M1' and 'M2' and oxide ions. Oxide ions form a ccp lattice structure. The cation 'M1' occupies 50% of octahedral voids and the cation 'M2' occupies 12.5% of tetrahedral voids of oxide lattice. The oxidation numbers of 'M1' and 'M2' are respectively:
NTA JEE Main 6th September 2020 Shift 2 - Question 34
A set of solutions is prepared using 180 g of water as a solvent and 10 g of different non-volatile solutes A, B and C. The relative lowering of vapour pressure in the presence of these solutes are in the order [Given, molar mass of $$A = 100\,\text{g mol}^{-1}$$; $$B = 200\,\text{g mol}^{-1}$$; $$C = 10,000\,\text{g mol}^{-1}$$]
NTA JEE Main 6th September 2020 Shift 2 - Question 35
For the given cell; $$\text{Cu}(s)|\text{Cu}^{2+}(C_1\,M)||\text{Cu}^{2+}(C_2\,M)|\text{Cu}(s)$$
change in Gibbs energy $$(\Delta G)$$ is negative, it:
NTA JEE Main 6th September 2020 Shift 2 - Question 36
The element that can be refined by distillation is:
NTA JEE Main 6th September 2020 Shift 2 - Question 37
The reaction of NO with $$\text{N}_2\text{O}_4$$ at 250 K gives:
NTA JEE Main 6th September 2020 Shift 2 - Question 38
Reaction of an inorganic sulphite X with dilute $$\text{H}_2\text{SO}_4$$ generated compound Y. Reaction of Y with NaOH gives X. Further, the reaction of X with Y and water affords compound Z. Y and X, respectively, are:
NTA JEE Main 6th September 2020 Shift 2 - Question 39
Mischmetal is an alloy consisting mainly of:
NTA JEE Main 6th September 2020 Shift 2 - Question 40
For a $$d^4$$ metal ion in an octahedral field, the correct electronic configuration is:


