NTA JEE Main 6th September 2020 Shift 2
For the following questions answer them individually
NTA JEE Main 6th September 2020 Shift 2 - Question 21
The centre of mass of a solid hemisphere of radius $$8\,\text{cm}$$ is $$x\,\text{cm}$$ from the centre of the flat surface. Then value of $$x$$ is___
789
456
123
0.-
Clear All
NTA JEE Main 6th September 2020 Shift 2 - Question 22
An engine operates by taking a monatomic ideal gas through the cycle shown in the figure. The percentage efficiency of the engine is close to___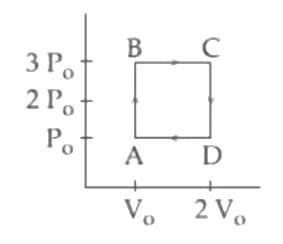
789
456
123
0.-
Clear All
NTA JEE Main 6th September 2020 Shift 2 - Question 23
In a series LR circuit, power of $$400\,\text{W}$$ is dissipated from a source of $$250\,\text{V}$$, $$50\,\text{Hz}$$. The power factor of the circuit is $$0.8$$. In order to bring the power factor to unity, a capacitor of value C is added in series to the L and R. Taking the value of C as $$\left(\frac{n}{3\pi}\right)\,\mu F$$, then value of n is___
789
456
123
0.-
Clear All
NTA JEE Main 6th September 2020 Shift 2 - Question 24
A Young's double-slit experiment is performed using monochromatic light of wavelength $$\lambda$$. The intensity of light at a point on the screen, where the path difference is $$\lambda$$, is $$K$$ units. The intensity of light at a point where the path difference is $$\dfrac{\lambda}{6}$$ is given by $$\dfrac{nK}{12}$$, where n is an integer. The value of n is___
789
456
123
0.-
Clear All
NTA JEE Main 6th September 2020 Shift 2 - Question 25
The output characteristics of a transistor is shown in the figure. When $$V_{CE}$$ is $$10\,\text{V}$$ and $$I_C = 4.0\,\text{mA}$$, then value of $$\beta_{ac}$$ is___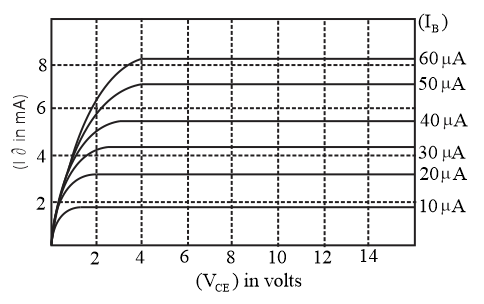
789
456
123
0.-
Clear All
NTA JEE Main 6th September 2020 Shift 2 - Question 26
The average molar mass of chlorine is $$35.5\,\text{g mol}^{-1}$$. The ratio of $$^{35}\text{Cl}$$ to $$^{37}\text{Cl}$$ in naturally occurring chlorine is close to:
NTA JEE Main 6th September 2020 Shift 2 - Question 27
For a reaction $$4M(s) + nO_2(g) \to 2M_2O_n(s)$$. The free energy change is plotted as a function of temperature. The temperature below which the oxide is stable could be inferred from the plot as the point at which:
NTA JEE Main 6th September 2020 Shift 2 - Question 28
The value of $$K_c$$ is 64 at 800 K for the reaction
$$\text{N}_2(g) + 3\text{H}_2(g) \to 2\text{NH}_3(g)$$
The value of $$K_c$$ for the following reaction is:
$$\text{NH}_3(g) \to \frac{1}{2}\text{N}_2(g) + \frac{3}{2}\text{H}_2(g)$$
NTA JEE Main 6th September 2020 Shift 2 - Question 29
Dihydrogen of high purity (> 99.95%) is obtained through:
NTA JEE Main 6th September 2020 Shift 2 - Question 30
Match the following compounds (Column-I) with their uses (Column-II):
S. No. Column-I S.No. Column-II
(I) $$\text{Ca(OH)}_2$$ - (A) casts of statues
(II) NaCl - (B) white wash
(III) $$\text{CaSO}_4 \cdot \frac{1}{2}\text{H}_2\text{O}$$ - (C) antacid
(IV) $$\text{CaCO}_3$$ - (D) washing soda preparation


