NTA JEE Main 5th September 2020 Shift 2
For the following questions answer them individually
NTA JEE Main 5th September 2020 Shift 2 - Question 31
Hydrogen peroxide, in the pure state, is:
NTA JEE Main 5th September 2020 Shift 2 - Question 32
The one that is NOT suitable for the removal of permanent hardness of water is:
NTA JEE Main 5th September 2020 Shift 2 - Question 33
Among the following compounds, geometrical isomerism is exhibited by:
NTA JEE Main 5th September 2020 Shift 2 - Question 34
The final major product of the following reaction is:
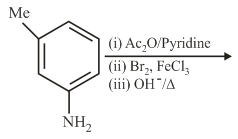
NTA JEE Main 5th September 2020 Shift 2 - Question 35
The major product formed in the following reaction is:
$$\text{CH}_2\text{CH} = \text{CHCH(CH}_3)_2 \xrightarrow{\text{HBr}}$$
NTA JEE Main 5th September 2020 Shift 2 - Question 36
An element crystallises in a face-centred cubic (fcc) unit cell with cell edge $$a$$. The distance between the centres of two nearest octahedral voids in the crystal lattice is:
NTA JEE Main 5th September 2020 Shift 2 - Question 37
The variation of molar conductivity with concentration of an electrolyte (X) in aqueous solution is shown in the given figure.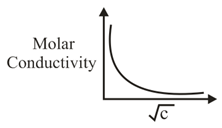
The electrolyte X is:
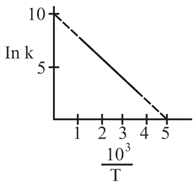
NTA JEE Main 5th September 2020 Shift 2 - Question 38
The rate constant $$(k)$$ of a reaction is measured at different temperature $$(T)$$, and the data are plotted in the given figure. The activation energy of the reaction in $$\text{kJ mol}^{-1}$$ is: (R is gas constant)
NTA JEE Main 5th September 2020 Shift 2 - Question 39
Adsorption of a gas follows Freundlich adsorption isotherm. If $$x$$ is the mass of the gas adsorbed on mass $$m$$ of the adsorbent, the correct plot of $$\frac{x}{m}$$ versus $$p$$ is:
NTA JEE Main 5th September 2020 Shift 2 - Question 40
Boron and silicon of very high purity can be obtained through:


