NTA JEE Main 5th September 2020 Shift 2
For the following questions answer them individually
NTA JEE Main 5th September 2020 Shift 2 - Question 41
Reaction of ammonia with excess $$Cl_2$$ gives:
NTA JEE Main 5th September 2020 Shift 2 - Question 42
Consider the complex ions, trans-$$[\text{Co(en)}_2\text{Cl}_2]^+$$ (A) and cis-$$[\text{Co(en)}_2\text{Cl}_2]^+$$ (B). The correct statement regarding them is:
NTA JEE Main 5th September 2020 Shift 2 - Question 43
The major product of the following reaction is:
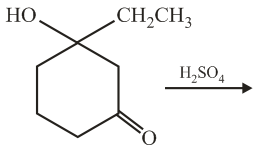
NTA JEE Main 5th September 2020 Shift 2 - Question 44
Which one of the following polymers is not obtained by condensation polymerisation?
NTA JEE Main 5th September 2020 Shift 2 - Question 45
The following molecule acts as an:
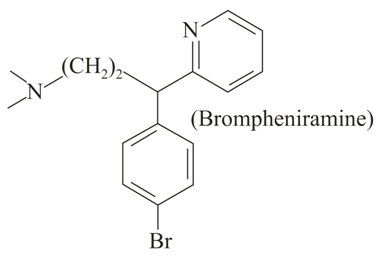
NTA JEE Main 5th September 2020 Shift 2 - Question 46
For a dimerization reaction,
$$2A(g) \to A_2(g)$$
at 298K, $$\Delta U^- = -20\,\text{kJ mol}^{-1}$$, $$\Delta S^- = -30\,\text{JK}^{-1}\text{mol}^{-1}$$, then the $$\Delta G^-$$ will be .....J.
789
456
123
0.-
Clear All
NTA JEE Main 5th September 2020 Shift 2 - Question 47
For a reaction $$X + Y = 2Z$$, $$1.0\,\text{mol}$$ of X, $$1.5\,\text{mol}$$ of Y and $$0.5\,\text{mol}$$ of Z were taken in a 1L vessel and allowed to react. At equilibrium, the concentration of Z was $$1.0\,\text{mol L}^{-1}$$. The equilibrium constant of the reaction is $$\frac{x}{15}$$. The value of x is.......
789
456
123
0.-
Clear All
NTA JEE Main 5th September 2020 Shift 2 - Question 48
The volume, in mL, of $$0.02\,\text{M}\,\text{K}_2\text{Cr}_2\text{O}_7$$ solution required to react with $$0.288\,\text{g}$$ of ferrous oxalate in acidic medium is............ (Molar mass of Fe = $$56\,\text{g mol}^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 5th September 2020 Shift 2 - Question 49
Considering that $$\Delta_0 > P$$, the magnetic moment (in BM) of $$[\text{Ru}(\text{H}_2\text{O})_6]^{2+}$$ would be
789
456
123
0.-
Clear All
NTA JEE Main 5th September 2020 Shift 2 - Question 50
The number of chiral carbons present in sucrose is..........
789
456
123
0.-
Clear All


