NTA JEE Main 5th September 2020 Shift 2
For the following questions answer them individually
NTA JEE Main 5th September 2020 Shift 2 - Question 21
A body of mass $$2\,\text{kg}$$ is driven by an engine delivering a constant power of $$1\,\text{J s}^{-1}$$. The body starts from rest and moves in a straight line. After $$9\,\text{s}$$, the body has moved a distance (in m)....
789
456
123
0.-
Clear All
NTA JEE Main 5th September 2020 Shift 2 - Question 22
A thin rod of mass $$0.9\,\text{kg}$$ and length $$1\,\text{m}$$ is suspended, at rest, from one end so that it can freely oscillate in the vertical plane. A particle of mass $$0.1\,\text{kg}$$ moving in a straight line with velocity $$80\,\text{m s}^{-1}$$ hits the rod at its bottom most point and sticks to it (see figure). The angular speed (in $$\text{rad s}^{-1}$$) of the rod immediately after the collision will be...........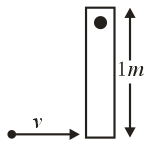
789
456
123
0.-
Clear All
NTA JEE Main 5th September 2020 Shift 2 - Question 23
Nitrogen gas is at $$300\,^\circ\text{C}$$ temperature. The temperature (in K) at which the rms speed of a $$\text{H}_2$$ molecule would be equal to the rms speed of a nitrogen molecule, is........... (Molar mass of $$\text{N}_2$$ gas 28 g).
789
456
123
0.-
Clear All
NTA JEE Main 5th September 2020 Shift 2 - Question 24
A prism of angle $$A = 1^\circ$$, $$\mu = 1.5$$. A good estimate for the minimum angle of deviation (in degrees) is close to $$\frac{N}{10}$$. Value of N is.........
789
456
123
0.-
Clear All
NTA JEE Main 5th September 2020 Shift 2 - Question 25
The surface of a metal is illuminated alternately with photons of energies $$E_1 = 4\,\text{eV}$$ and $$E_2 = 2.5\,\text{eV}$$ respectively. The ratio of maximum speeds of the photoelectrons emitted in the two cases is 2. The work function of the metal in (eV) is..........
789
456
123
0.-
Clear All
NTA JEE Main 5th September 2020 Shift 2 - Question 26
The correct statement about probability density (except at infinite distance from nucleus) is:
NTA JEE Main 5th September 2020 Shift 2 - Question 27
The correct order of the ionic radii of $$\text{O}^{2-}$$, $$\text{N}^{3-}$$, $$\text{F}^-$$, $$\text{Mg}^{2+}$$, $$\text{Na}^+$$ and $$\text{Al}^{3+}$$ is:
NTA JEE Main 5th September 2020 Shift 2 - Question 28
The increasing order of boiling points of the following compounds is: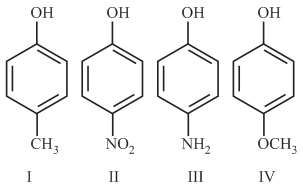
NTA JEE Main 5th September 2020 Shift 2 - Question 29
The compound that has the largest $$H-M-H$$ bond angle ($$M = N, O, S, C$$) is:
NTA JEE Main 5th September 2020 Shift 2 - Question 30
Lattice enthalpy and enthalpy of solution of NaCl are $$788\,\text{kJ mol}^{-1}$$ and $$4\,\text{kJ mol}^{-1}$$, respectively. The hydration enthalpy of $$NaCl$$ is:


