NTA JEE Main 30th January 2023 Shift 2
For the following questions answer them individually
NTA JEE Main 30th January 2023 Shift 2 - Question 41
The water quality of a pond was analysed and its BOD was found to be 4. The pond has
NTA JEE Main 30th January 2023 Shift 2 - Question 42
Given below are two statements:
Statement I: During Electrolytic refining, the pure metal is made to act as anode and its impure metallic form is used as cathode.
Statement II: During the Hall-Heroult electrolysis process, purified Al$$_2$$O$$_3$$ is mixed with Na$$_3$$AlF$$_6$$ to lower the melting point of the mixture.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 30th January 2023 Shift 2 - Question 43
Formulae for Nessler's reagent is:
NTA JEE Main 30th January 2023 Shift 2 - Question 44
KMnO$$_4$$ oxidises I$$^-$$ in acidic and neutral/faintly alkaline solution, respectively to
NTA JEE Main 30th January 2023 Shift 2 - Question 45
Match List I with List II:
| List I (Complexes) | List II (Hybridisation) |
|---|---|
| (A) [Ni(CO)$$_4$$] | I. sp$$^3$$ |
| (B) [Cu(NH$$_3$$)$$_4$$]$$^{2+}$$ | II. dsp$$^2$$ |
| (C) [Fe(NH$$_3$$)$$_6$$]$$^{2+}$$ | III. sp$$^3$$d$$^2$$ |
| (D) [Fe(H$$_2$$O)$$_6$$]$$^{2+}$$ | IV. d$$^2$$sp$$^3$$ |
NTA JEE Main 30th January 2023 Shift 2 - Question 46
1 L, 0.02M solution of [Co(NH$$_3$$)$$_5$$ SO$$_4$$]Br is mixed with 1 L, 0.02M solution of [Co(NH$$_3$$)$$_5$$Br]SO$$_4$$. The resulting solution is divided into two equal parts (X) and treated with excess AgNO$$_3$$ solution and BaCl$$_2$$ solution respectively as shown below:
1 L Solution (X) + AgNO$$_3$$ solution (excess) $$\to$$ Y
1 L Solution (X) + BaCl$$_2$$ solution (excess) $$\to$$ Z
The number of moles of Y and Z respectively are
NTA JEE Main 30th January 2023 Shift 2 - Question 47
The Cl $$-$$ Co $$-$$ Cl bond angle values in a fac-[Co(NH$$_3$$)$$_3$$Cl$$_3$$] complex is/are:
NTA JEE Main 30th January 2023 Shift 2 - Question 48
Decreasing order towards S$$_N$$1 reaction for the following compounds is:
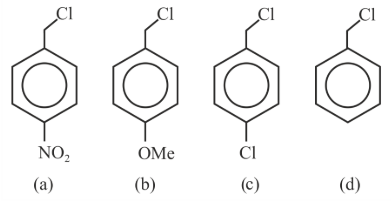
NTA JEE Main 30th January 2023 Shift 2 - Question 49
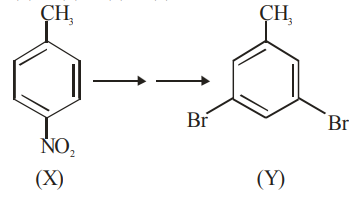
In the above conversion of compound (X) to product (Y), the sequence of reagents to be used will be:
NTA JEE Main 30th January 2023 Shift 2 - Question 50
Given below are two statements: One is labelled as Assertion A and the other labelled as Reason R.
Assertion A: Antihistamines do not affect the secretion of acid in stomach.
Reason R: Antiallergic and antacid drugs work on different receptors.
In the light of the above statements, choose the correct answer from the options given below:


