NTA JEE Main 30th January 2023 Shift 2
For the following questions answer them individually
NTA JEE Main 30th January 2023 Shift 2 - Question 31
The wave function $$(\Psi)$$ of $$2s$$ is given by
$$\Psi_{2s} = \frac{1}{2\sqrt{2\pi}}\left(\frac{1}{a_0}\right)^{1/2}\left(2 - \frac{r}{a_0}\right)e^{-r/2a_0}$$
At $$r = r_0$$, radial node is formed. Thus, $$r_0$$ in terms of $$a_0$$
NTA JEE Main 30th January 2023 Shift 2 - Question 32
Maximum number of electrons that can be accommodated in shell with $$n = 4$$ are:
NTA JEE Main 30th January 2023 Shift 2 - Question 33
Bond dissociation energy of E $$-$$ H bond of the "H$$_2$$E" hydrides of group 16 elements (given below), follows order.
(A) O
(B) S
(C) Se
(D) Te
NTA JEE Main 30th January 2023 Shift 2 - Question 34
Chlorides of which metal are soluble in organic solvents:
NTA JEE Main 30th January 2023 Shift 2 - Question 35
Which of the following reaction is correct?
NTA JEE Main 30th January 2023 Shift 2 - Question 36
Boric acid in solid, whereas BF$$_3$$ is gas at room temperature because of
NTA JEE Main 30th January 2023 Shift 2 - Question 37
The most stable carbocation for the following is:
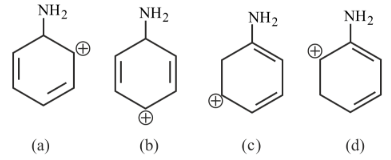
NTA JEE Main 30th January 2023 Shift 2 - Question 38
The correct order of pK$$_a$$ values for the following compounds is:
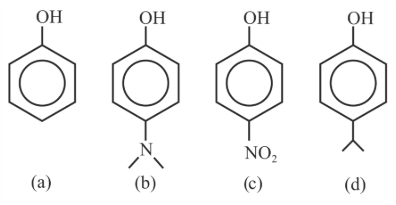
NTA JEE Main 30th January 2023 Shift 2 - Question 39
Match List I with List II:
| List I (Mixture) | List II (Separation Technique) |
|---|---|
| (A) CHCl$$_3$$ + C$$_6$$H$$_5$$NH$$_2$$ | I. Steam distillation |
| (B) C$$_6$$H$$_{14}$$ + C$$_5$$H$$_{12}$$ | II. Differential extraction |
| (C) C$$_6$$H$$_5$$NH$$_2$$ + H$$_2$$O | III. Distillation |
| (D) Organic compound in H$$_2$$O | IV. Fractional distillation |
NTA JEE Main 30th January 2023 Shift 2 - Question 40
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A:
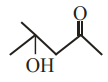
can be easily reduced using Zn $$-$$ Hg/HCl to
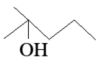
.
Reason R: Zn $$-$$ Hg/HCl is used to reduce carbonyl group to $$-$$CH$$_2-$$ group.
In the light of the above statements, choose the correct answer from the options given below:


