NTA JEE Main 30th January 2023 Shift 2
For the following questions answer them individually
NTA JEE Main 30th January 2023 Shift 2 - Question 51
1 mole of ideal gas is allowed to expand reversibly and adiabatically from a temperature of $$27°$$C. The work done is $$3$$ kJ mol$$^{-1}$$. The final temperature of the gas is ______ K (Nearest integer). Given $$C_v = 20$$ J mol$$^{-1}$$ K$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 30th January 2023 Shift 2 - Question 52
Consider the following equation:
$$2\text{SO}_2(g) + \text{O}_2(g) \rightleftharpoons 2\text{SO}_3(g), \Delta H = -190$$ kJ.
The number of factors which will increase the yield of SO$$_3$$ at equilibrium from the following is ______.
A. Increasing temperature
B. Increasing pressure
C. Adding more SO$$_2$$
D. Adding more O$$_2$$
E. Addition of catalyst
789
456
123
0.-
Clear All
NTA JEE Main 30th January 2023 Shift 2 - Question 53
The strength of $$50$$ volume solution of hydrogen peroxide is ______ g/L (Nearest integer).
Given: Molar mass of H$$_2$$O$$_2$$ is 34 g mol$$^{-1}$$. Molar volume of gas at STP = 22.7 L.
789
456
123
0.-
Clear All
NTA JEE Main 30th January 2023 Shift 2 - Question 54
Iron oxide FeO, crystallises in a cubic lattice with a unit cell edge length of $$5.0$$ $$\text{\AA}$$. If density of the FeO in the crystal is $$4.0$$ g cm$$^{-3}$$, then the number of FeO units present per unit cell is ______ (Nearest integer)
Given: Molar mass of Fe and O is 56 and 16 g mol$$^{-1}$$ respectively.
N$$_A = 6.0 \times 10^{23}$$ mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 30th January 2023 Shift 2 - Question 55
Lead storage battery contains $$38\%$$ by weight solution of H$$_2$$SO$$_4$$. The van't Hoff factor is $$2.67$$ at this concentration. The temperature in Kelvin at which the solution in the battery will freeze is ______ (Nearest integer).
Given K$$_f = 1.8$$ K kg mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 30th January 2023 Shift 2 - Question 56
The electrode potential of the following half cell at 298 K
X|X$$^{2+}$$(0.001M)||Y$$^{2+}$$(0.01M)|Y is ______ $$\times 10^{-2}$$ V (Nearest integer)
Given: E$$^0_{X^{2+}/X} = -2.36$$ V
E$$^0_{Y^{2+}/Y} = +0.36$$ V
$$\frac{2.303 RT}{F} = 0.06$$ V
789
456
123
0.-
Clear All
NTA JEE Main 30th January 2023 Shift 2 - Question 57
An organic compound undergoes first order decomposition. If the time taken for the $$60\%$$ decomposition is $$540$$ s, then the time required for $$90\%$$ decomposition will be ______ s. (Nearest integer).
Given: $$\ln 10 = 2.3$$; $$\log 2 = 0.3$$
789
456
123
0.-
Clear All
NTA JEE Main 30th January 2023 Shift 2 - Question 58
The graph of $$\log \frac{x}{m}$$ vs $$\log p$$ for an adsorption process is a straight line inclined at an angle of $$45°$$ with intercept equal to $$0.6020$$. The mass of gas adsorbed per unit mass of adsorbent at the pressure of $$0.4$$ atm is ______ $$\times 10^{-1}$$ (Nearest integer)
Given: $$\log 2 = 0.3010$$
789
456
123
0.-
Clear All
NTA JEE Main 30th January 2023 Shift 2 - Question 59
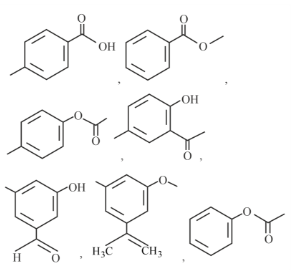
Number of compounds from the following which will not dissolve in cold NaHCO$$_3$$ and NaOH solutions but will dissolve in hot NaOH solution is ______.
789
456
123
0.-
Clear All
NTA JEE Main 30th January 2023 Shift 2 - Question 60
A short peptide on complete hydrolysis produces 3 moles of glycine (G), two moles of leucine (L) and two moles of valine (V) per mole of peptide. The number of peptide linkages in it are ______.
789
456
123
0.-
Clear All


