NTA JEE Main 30th January 2023 Shift 1
For the following questions answer them individually
NTA JEE Main 30th January 2023 Shift 1 - Question 1
Electric field in a certain region is given by $$\vec{E} = \frac{A}{x^2}\hat{i} + \frac{B}{y^3}\hat{j}$$. The SI unit of $$A$$ and $$B$$ are:
NTA JEE Main 30th January 2023 Shift 1 - Question 2
Match Column-I with Column-II:
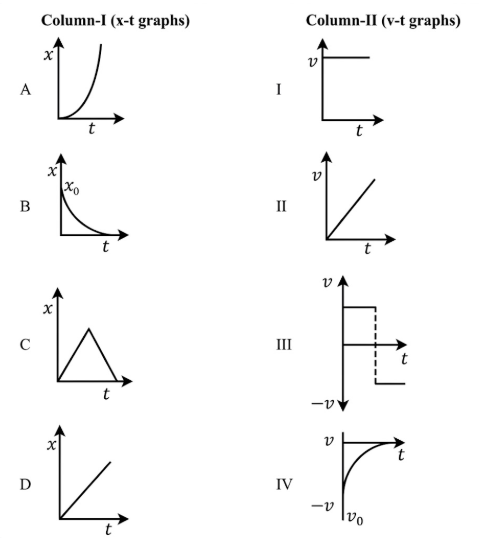
Choose the correct answer from the options given below:
NTA JEE Main 30th January 2023 Shift 1 - Question 3
The figure represents the momentum time $$(p - t)$$ curve for a particle moving along an axis under the influence of the force. Identify the regions on the graph where the magnitude of the force is maximum and minimum respectively?
If $$t_3 - t_2 < t_1$$
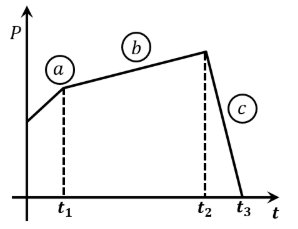
NTA JEE Main 30th January 2023 Shift 1 - Question 4
As per the given figure, a small ball $$P$$ slides down the quadrant of a circle and hits the other ball $$Q$$ of equal mass which is initially at rest. Neglecting the effect of friction and assume the collision to be elastic, the velocity of ball $$Q$$ after collision will be: ($$g = 10$$ m s$$^{-2}$$)
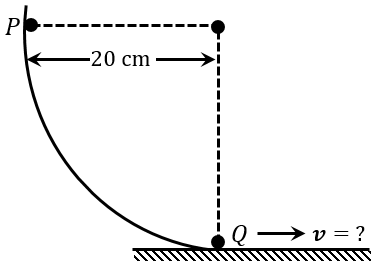
NTA JEE Main 30th January 2023 Shift 1 - Question 5
A ball of mass $$200$$ g rests on a vertical post of height $$20$$ m. A bullet of mass $$10$$ g, travelling in horizontal direction, hits the centre of the ball. After collision both travels independently. The ball hits the ground at a distance $$30$$ m and the bullet at a distance of $$120$$ m from the foot of the post. The value of initial velocity of the bullet will be (if $$g = 10$$ m s$$^{-2}$$):
NTA JEE Main 30th January 2023 Shift 1 - Question 6
If the gravitational field in the space is given as $$-\frac{K}{r^2}$$. Taking the reference point to be at $$r = 2$$ cm with gravitational potential $$V = 10$$ J kg$$^{-1}$$. Find the gravitational potentials at $$r = 3$$ cm in SI unit (Given, that $$K = 6$$ J cm kg$$^{-1}$$)
NTA JEE Main 30th January 2023 Shift 1 - Question 7
Choose the correct relationship between Poisson ratio $$\sigma$$, bulk modulus $$(K)$$ and modulus of rigidity $$\eta$$ of a given solid object:
NTA JEE Main 30th January 2023 Shift 1 - Question 8
The height of liquid column raised in a capillary tube of certain radius when dipped in liquid $$A$$ vertically is, $$5$$ cm. If the tube is dipped in a similar manner in another liquid $$B$$ of surface tension and density double the values of liquid $$A$$, the height of liquid column raised in liquid B would be ______ m.
NTA JEE Main 30th January 2023 Shift 1 - Question 9
Heat is given to an ideal gas in an isothermal process.
A. Internal energy of the gas will decrease.
B. Internal energy of the gas will increase.
C. Internal energy of the gas will not change.
D. The gas will do positive work.
E. The gas will do negative work.
Choose the correct answer from the options given below:
NTA JEE Main 30th January 2023 Shift 1 - Question 10
The pressure $$(P)$$ and temperature $$(T)$$ relationship of an ideal gas obeys the equation $$PT^2 =$$ constant. The volume expansion coefficient of the gas will be:


