NTA JEE Main 3rd September 2020 Shift 2
For the following questions answer them individually
NTA JEE Main 3rd September 2020 Shift 2 - Question 31
100 mL of 0.1 M HCl is taken in a beaker and to it 100 mL 0.1 M NaOH of is added in steps of 2 mL and the pH is continuously measured. Which of the following graphs correctly depicts the change in pH?
NTA JEE Main 3rd September 2020 Shift 2 - Question 32
Among the statements (I - IV), the correct ones are:
(I) Be has smaller atomic radius compared to Mg.
(II) Be has higher ionization enthalpy than Al.
(III) Charge/radius ratio of Be is greater than that of Al.
(IV) Both Be and Al form mainly covalent compounds
NTA JEE Main 3rd September 2020 Shift 2 - Question 33
The incorrect statement(s) among (a) - (d) regarding acid rain is (are):
(a) It can corrode water pipes
(b) It can damage structures made up of stone.
(c) It cannot cause respiratory ailments in animals
(d) It is not harmful for trees
NTA JEE Main 3rd September 2020 Shift 2 - Question 34
For the reaction $$2A + 3B + \frac{3}{2}C \to 3P$$, which statement is correct?
NTA JEE Main 3rd September 2020 Shift 2 - Question 35
The incorrect statement is:
NTA JEE Main 3rd September 2020 Shift 2 - Question 36
Complex A has a composition of $$H_{12}O_6Cl_3Cr$$. If the complex on treatment with conc. $$H_2SO_4$$ loses 13.5% of its original mass, the correct molecular formula of A is:
[Given: atomic mass of Cr = 52 amu and Cl = 35 amu]
NTA JEE Main 3rd September 2020 Shift 2 - Question 37
The d-electron configuration of $$[Ru(en)_3]Cl_2$$, and $$[Fe(H_2O)_6]Cl_2$$ respectively are:
NTA JEE Main 3rd September 2020 Shift 2 - Question 38
The decreasing order of reactivity of the following compounds towards nucleophilic substitution ($$S_N2$$) is:
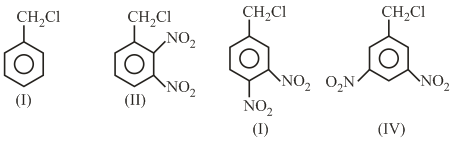
NTA JEE Main 3rd September 2020 Shift 2 - Question 39
The major product in the following reaction is:
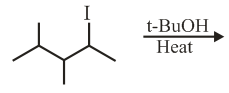
NTA JEE Main 3rd September 2020 Shift 2 - Question 40
Consider the following reaction:
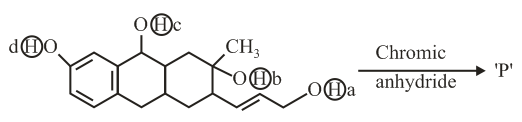
The product 'P' gives positive ceric ammonium nitrate test. This is because of the presence of which of these -OH group(s)?


