NTA JEE Main 3rd September 2020 Shift 2
For the following questions answer them individually
NTA JEE Main 3rd September 2020 Shift 2 - Question 41
The increasing order of the reactivity of the following compounds in nucleophile addition reaction is:
Propanal, Benzaldehyde, Propanone, Butanone
NTA JEE Main 3rd September 2020 Shift 2 - Question 42
The compound A in the following reactions is:
$$A \xrightarrow[(ii)Conc. H_{2}SO_{4}/ \Delta]{(i)CH_{3}MgBr/H_{2}O} B \xrightarrow[(ii) Zn/H_{2}O]{(i) O_{3}} C + D$$
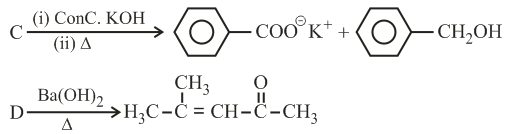
NTA JEE Main 3rd September 2020 Shift 2 - Question 43
Three isomers A, B and C (mol. formula $$C_8H_{11}N$$) give the following results:
A and C $$\xrightarrow{Diazotization}$$ P + Q $$\xrightarrow[(ii) acidation(KMnO_4+H^+)]{(i) Hydrolysis}$$ R (product of A) + S (product of C)
R has lower boiling point than S
B $$\xrightarrow{C_6H_5SO_2Cl}$$ alkali-insoluble product
A, B and C, respectively are:
NTA JEE Main 3rd September 2020 Shift 2 - Question 44
An ionic micelle is formed on the addition of:
NTA JEE Main 3rd September 2020 Shift 2 - Question 45
Match the following drugs with their therapeutic actions:
(i) Ranitidine (a) Antidepressant
(ii) Nardil (Phenelzine) (b) Antibiotic
(iii) Chloramphenicol (c) Antihistamine
(iv) Dimetane (Brompheniramine) (d) Antacid
(e) Analgesic
NTA JEE Main 3rd September 2020 Shift 2 - Question 46
$$0.023 \times 10^{22}$$ molecules are present in 10g of a substance 'x'. The molarity of a solution containing 5g of substance 'x' in 2 L solution is _________ $$\times 10^{-3}$$
789
456
123
0.-
Clear All
NTA JEE Main 3rd September 2020 Shift 2 - Question 47
If 250 cm$$^3$$ of an aqueous solution containing 0.73g of a protein A is isotonic with one litre of another aqueous solution containing 1.65g of a protein B, at 298K, the ratio of the molecular masses of A and B is _________ $$\times 10^{-2}$$ (to the nearest integer).
789
456
123
0.-
Clear All
NTA JEE Main 3rd September 2020 Shift 2 - Question 48
An acidic solution of dichromate is electrolyzed for 8 minutes using 2 A current. As per the following equation $$Cr_2O_7^{2-} + 14H^+ + 6e^- \to 2Cr^{3+} + 7H_2O$$
The amount of $$Cr^{3+}$$ obtained was 0.104g. The efficiency of the process (in %) is (Take: F = 960000C, At. mass of chromium = 52)
789
456
123
0.-
Clear All
NTA JEE Main 3rd September 2020 Shift 2 - Question 49
The volume (in mL) of 0.1 N NaOH required to neutralise 10 mL of 0.1 N phosphinic acid is __________.
789
456
123
0.-
Clear All
NTA JEE Main 3rd September 2020 Shift 2 - Question 50
The number of
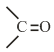
groups present in a tripeptide Asp - Glu - Lys is ____
789
456
123
0.-
Clear All


