NTA JEE Main 3rd September 2020 Shift 2
For the following questions answer them individually
NTA JEE Main 3rd September 2020 Shift 2 - Question 21
A block starts moving up an inclined plane of inclination 30° with an initial velocity of $$v_0$$. It comes back to its initial position with velocity $$\frac{v_0}{2}$$. The value of the coefficient of kinetic friction between the block and the inclined plane is close to $$\frac{I}{1000}$$. The nearest integer to I is:
789
456
123
0.-
Clear All
NTA JEE Main 3rd September 2020 Shift 2 - Question 22
An massless equilateral triangle EFG of side 'a' (As shown in figure) has three particles of mass m situated at its vertices. The moment of inertia of the system about the line EX perpendicular to EG in the plane of EFG is $$\frac{N}{20}ma^2$$ where N is an integer. The value of N is __________.
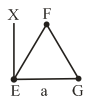
789
456
123
0.-
Clear All
NTA JEE Main 3rd September 2020 Shift 2 - Question 23
If minimum possible work is done by a refrigerator in converting 100 grams of water at 0°C to ice, how much heat (in calories) is released to the surroundings at temperature 27°C (Latent heat of ice = 80 Cal/gram) to the nearest integer?
789
456
123
0.-
Clear All
NTA JEE Main 3rd September 2020 Shift 2 - Question 24
A galvanometer coil has 500 turns and each turn has an average area of $$3 \times 10^{-4}$$ m$$^2$$. If a torque of 1.5 Nm is required to keep this coil parallel to a magnetic field when a current of 0.5A is flowing through it, the strength of the field (in T) is __________.
789
456
123
0.-
Clear All
NTA JEE Main 3rd September 2020 Shift 2 - Question 25
When an object is kept at a distance of 30 cm from a concave mirror, the image is formed at a distance of 10 cm from the mirror. If the object is moved with a speed of 9 cm s$$^{-1}$$, the speed (in cm s$$^{-1}$$) with which image moves at that instant is
789
456
123
0.-
Clear All
NTA JEE Main 3rd September 2020 Shift 2 - Question 26
The strengths of 5.6 volume hydrogen peroxide (of density 1 g/mL) in terms of mass percentage and molarity(M) respectively, are: (Take molar mass of hydrogen peroxide as 34 g/mol)
NTA JEE Main 3rd September 2020 Shift 2 - Question 27
Consider the hypothetical situation where the azimuthal quantum number, $$\ell$$, takes values 0, 1, 2, ......... n + 1. Where n is the principal quantum number. Then, the element with atomic number:
NTA JEE Main 3rd September 2020 Shift 2 - Question 28
The five successive ionization enthalpies of an element are 800, 2427, 3658, 35024, 32824 kJ mol$$^{-1}$$. The number of valence electrons in the element is:
NTA JEE Main 3rd September 2020 Shift 2 - Question 29
Consider the following molecules and statements related to them:
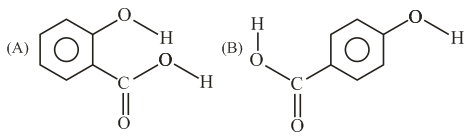
(a) (B) is more likely to be crystalline than (A)
(b) (B) has higher boiling point than (A)
(c) (B) dissolves more readily than (A) in water
Identify the correct option from below:
NTA JEE Main 3rd September 2020 Shift 2 - Question 30
A mixture of one mole each of $$H_2$$, He and $$O_2$$ each are enclosed in a cylinder of volume V at temperature T. If the partial pressure of $$H_2$$ is 2 atm, the total pressure of the gases in the cylinder is:


