NTA JEE Main 27th July 2022 Shift 1
For the following questions answer them individually
NTA JEE Main 27th July 2022 Shift 1 - Question 41
Boiling point of a $$2\%$$ aqueous solution of a nonvolatile solute A is equal to the boiling point of $$8\%$$ aqueous solution of a non-volatile solute B. The relation between molecular weights of A and B is
NTA JEE Main 27th July 2022 Shift 1 - Question 42
Which of the following methods are not used to refine any metal?
(A) Liquation
(B) Calcination
(C) Electrolysis
(D) Leaching
(E) Distillation
Choose the correct answer from the options given below
NTA JEE Main 27th July 2022 Shift 1 - Question 43
Which oxoacid of phosphorous has the highest number of oxygen atoms present in its chemical formula?
NTA JEE Main 27th July 2022 Shift 1 - Question 44
Given below are two statements:
Statement I: Iron (III) catalyst, acidified $$K_2Cr_2O_7$$ and neutral $$KMnO_4$$ have the ability to oxidise $$I^-$$ to $$I_2$$ independently.
Statement II: Manganate ion is paramagnetic in nature and involves $$p\pi - p\pi$$ bonding.
In the light of the above statements, choose the correct answer from the options.
NTA JEE Main 27th July 2022 Shift 1 - Question 45
The total number of $$Mn = O$$ bonds in $$Mn_2O_7$$ is
NTA JEE Main 27th July 2022 Shift 1 - Question 46
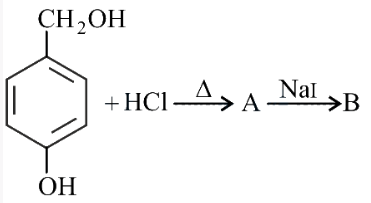
In the above reaction, product B is
NTA JEE Main 27th July 2022 Shift 1 - Question 47
Match List-I with List-II

Choose the correct answer from the options given below
NTA JEE Main 27th July 2022 Shift 1 - Question 48
Match List I with List II

Choose the correct answer from the options given below
NTA JEE Main 27th July 2022 Shift 1 - Question 49
Match List-I with List-II.
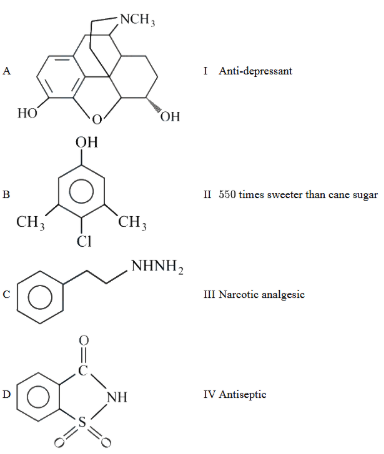
Choose the correct answer from the options given below
NTA JEE Main 27th July 2022 Shift 1 - Question 50
A sugar 'X' dehydrates very slowly under acidic condition to give furfural which on further reaction with resorcinol gives the coloured product after sometime. Sugar 'X' is


