NTA JEE Main 27th July 2022 Shift 1
For the following questions answer them individually
NTA JEE Main 27th July 2022 Shift 1 - Question 31
$$250 \text{ g}$$ solution of D-glucose in water contains $$10.8\%$$ of carbon by weight. The molality of the solution is nearest to (Given: Atomic Weights are $$H = 1u; C = 12u; O = 16u$$)
NTA JEE Main 27th July 2022 Shift 1 - Question 32
Given below are two statements. One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Energy of $$2s$$ orbital of hydrogen atom is greater than that of $$2s$$ orbital of lithium.
Reason R: Energies of the orbitals in the same subshell decrease with increase in the atomic number.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 27th July 2022 Shift 1 - Question 33
The incorrect statement is
NTA JEE Main 27th July 2022 Shift 1 - Question 34
Given below are two statements.
Statement I: $$O_2$$, $$Cu^{2+}$$ and $$Fe^{3+}$$ are weakly attracted by magnetic field and are magnetized in the same direction as magnetic field.
Statement II: $$NaCl$$ and $$H_2O$$ are weakly magnetized in opposite direction to magnetic field.
In the light of the above statements, choose the most appropriate answer form the options given below
NTA JEE Main 27th July 2022 Shift 1 - Question 35
Given below are two statements. One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Activated charcoal adsorbs $$SO_2$$ more efficiently than $$CH_4$$.
Reason R: Gases with lower critical temperatures are readily adsorbed by activated charcoal.
In the light of the above statements, choose the correct answer from the options given below.
NTA JEE Main 27th July 2022 Shift 1 - Question 36
Given below are two statements:
Statement I: Hydrogen peroxide can act as an oxidizing agent in both acidic and basic conditions.
Statement II: Density of hydrogen peroxide at $$298 \text{ K}$$ is lower than that of $$D_2O$$.
In the light of the above statements. Choose the correct answer from the options
NTA JEE Main 27th July 2022 Shift 1 - Question 37
Given below are two statements
Statement I: The chlorides of Be and Al have Cl-bridged structure. Both are soluble in organic solvents and act as Lewis bases.
Statement II: Hydroxides of Be and Al dissolve in excess alkali to give beryllate and aluminate ions.
In the light of the above statements. Choose the correct answer from the options
NTA JEE Main 27th July 2022 Shift 1 - Question 38
Given below are two statements. One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: [6] Annulene, [8] Annulene and cis-[10] Annulene are respectively aromatic, not-aromatic and aromatic.
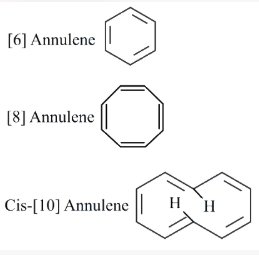
Reason R: Planarity is one of the requirements of aromatic systems.
In the light of the above statements, choose the most appropriate answer from the options given below.
NTA JEE Main 27th July 2022 Shift 1 - Question 39
In Carius method of estimation of halogen, $$0.45 \text{ g}$$ of an organic compound gave $$0.36 \text{ g}$$ of $$AgBr$$. Find out the percentage of bromine in the compound. (Molar masses: $$AgBr = 188 \text{ g mol}^{-1}; Br = 80 \text{ g mol}^{-1}$$)
NTA JEE Main 27th July 2022 Shift 1 - Question 40
Match List I with List II

Choose the correct answer from the options given below


