NTA JEE Main 27th July 2022 Shift 1
For the following questions answer them individually
NTA JEE Main 27th July 2022 Shift 1 - Question 51
Amongst the following the number of oxide(s) which are paramagnetic in nature is ______
$$Na_2O, KO_2, NO_2, N_2O, ClO_2, NO, SO_2, Cl_2O$$
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 1 - Question 52
According to MO theory, number of species/ions from the following having identical bond order is ______
$$CN^-, NO^+, O_2, O_2^+, O_2^{2+}$$
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 1 - Question 53
The molar heat capacity for an ideal gas at constant pressure is $$20.785 \text{ J K}^{-1} \text{ mol}^{-1}$$. The change in internal energy is $$5000 \text{ J}$$ upon heating it from $$300 \text{ K}$$ to $$500 \text{ K}$$. The number of moles of the gas at constant volume is ______ (Given: $$R = 8.314 \text{ J K}^{-1} \text{ mol}^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 1 - Question 54
At $$310 \text{ K}$$, the solubility of $$CaF_2$$ in water is $$2.34 \times 10^{-3} \text{ g/100 mL}$$. The solubility product of $$CaF_2$$ is ______ $$\times 10^{-8} (\text{mol/L})^3$$ (nearest integer). (Given molar mass: $$CaF_2 = 78 \text{ g mol}^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 1 - Question 55
$$20 \text{ mL}$$ of $$0.02M K_2Cr_2O_7$$ solution is used for the titration of $$10 \text{ mL}$$ of $$Fe^{2+}$$ solution in the acidic medium. The molarity of $$Fe^{2+}$$ solution is ______ $$\times 10^{-2} M$$
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 1 - Question 56
In the following reaction,
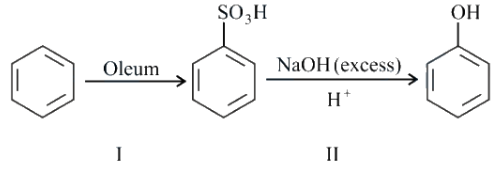
the $$\%$$ yield for reaction I is $$60\%$$ and that of reaction II is $$50\%$$. The overall yield of the complete reaction is ______ $$\%$$.
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 1 - Question 57
$$2NO + 2H_2 \rightarrow N_2 + 2H_2O$$. The above reaction has been studied at $$800°C$$.The related data are given in the table below

The order of the reaction with respect to NO is ______.
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 1 - Question 58
In the titration of $$KMnO_4$$ and oxalic acid in acidic medium, the change in oxidation number of carbon at the end point is ______.
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 1 - Question 59
The conductivity of a solution of complex with formula $$CoCl_3(NH_3)_4$$ corresponds to $$1:1$$ electrolyte, then the primary valency of central metal ion is ______.
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 1 - Question 60
Optical activity of an enantiomeric mixture is $$+12.6°$$ and the specific rotation of $$(+)$$ isomer is $$+30°$$. The optical purity is ______ $$\%$$.
789
456
123
0.-
Clear All


