NTA JEE Main 26th February 2021 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 31
The correct order of electron gain enthalpy is:
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 32
Which pair of oxides is acidic in nature?
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 33
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: In $$TlI_3$$, isomorphous to $$CsI_3$$, the metal is present in +1 oxidation state.
Reason R: Tl metal has fourteen f electrons in its electronic configuration.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 34
Match List-I with List-II.
| List-I (Molecule) | List-II (Bond order) |
|---|---|
| (a) $$Ne_2$$ | (i) 1 |
| (b) $$N_2$$ | (ii) 2 |
| (c) $$F_2$$ | (iii) 0 |
| (d) $$O_2$$ | (iv) 3 |
Choose the correct answer from the options given below:
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 35
Calgon is used for water treatment. Which of the following statement is NOT true about Calgon?
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 36
Which of the following forms of hydrogen emits low energy $$\beta^-$$ particles?
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 37
In
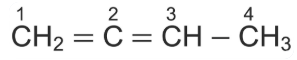
molecule, the hybridization of carbon 1, 2, 3 and 4 respectively, are:
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 38
The nature of charge on resulting colloidal particles when $$FeCl_3$$ is added to excess of hot water is:
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 39
Match List-I with List-II.
| List-I | List-II |
|---|---|
| (a) Sodium Carbonate | (i) Deacon |
| (b) Titanium | (ii) Castner-Kellner |
| (c) Chlorine | (iii) van-Arkel |
| (d) Sodium hydroxide | (iv) Solvay |
Choose the correct answer from the options given below:
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 40
Match List-I with List-II.
| List-I (Ore) | List-II (Metal) |
|---|---|
| (a) Siderite | (i) Cu |
| (b) Calamine | (ii) Ca |
| (c) Malachite | (iii) Fe |
| (d) Cryolite | (iv) Al |
| (v) Zn |
Choose the correct answer from the options given below:
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 41
Match List-I with List-II.
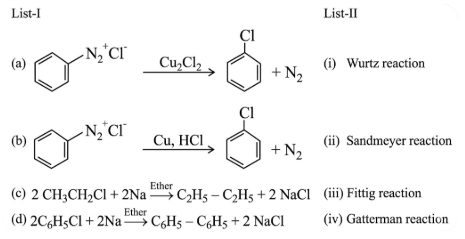
Choose the correct answer from the options given below :
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 42
Identify A in the given reaction.
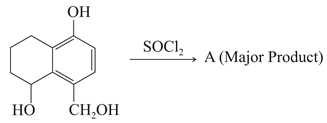
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 43
Considering the above reaction, the major product among the following is:
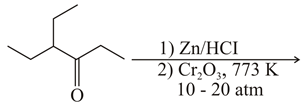
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 44
2, 4-DNP test can be used to identify:
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 45
Identify A in the given chemical reaction.
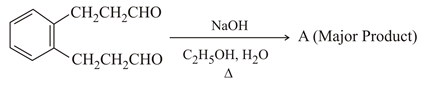
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 46
Identify A in the following chemical reaction.
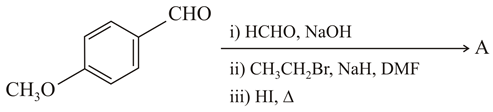
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 47
Ceric ammonium nitrate and $$CHCl_3$$ / alc. KOH are used for the identification of functional groups present in ______ and ______ respectively.
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 48
A. Phenyl methanamine
B. N, N-Dimethylaniline
C. N-Methyl aniline
D. Benzenamine
Choose the correct order of basic nature of the above amines.
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 49
Match List-I with List-II.
| List-I | List-II |
|---|---|
| (a) Sucrose | (i) $$\beta$$-D-Galactose and $$\beta$$-D-Glucose |
| (b) Lactose | (ii) $$\alpha$$-D-Glucose and $$\beta$$-D-Fructose |
| (c) Maltose | (iii) $$\alpha$$-D-Glucose and $$\alpha$$-D-Glucose |
Choose the correct answer from the options given below:
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 50
Seliwanoff test and Xanthoproteic test are used for the identification of ______ and ______ respectively.
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 51
The $$NaNO_3$$ weighed out to make 50 mL of an aqueous solution containing 70.0 mg Na$$^+$$ per mL is ______ g.
(Rounded off to the nearest integer) [Given: Atomic weight in g mol$$^{-1}$$ - Na: 23; N: 14; O: 16]
789
456
123
0.-
Clear All
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 52
A ball weighing 10 g is moving with a velocity of 90 m s$$^{-1}$$. If the uncertainty in its velocity is 5%, then the uncertainty in its position is ______ $$\times 10^{-33}$$ m. (Rounded off to the nearest integer)
[Given: h = $$6.63 \times 10^{-34}$$ Js]
789
456
123
0.-
Clear All
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 53
The average S - F bond energy in kJ mol$$^{-1}$$ of $$SF_6$$ is ______. (Rounded off to the nearest integer)
[Given: The values of standard enthalpy of formation of $$SF_6(g)$$, $$S(g)$$ and $$F(g)$$ are $$-1100$$, 275 and 80 kJ mol$$^{-1}$$ respectively.]
789
456
123
0.-
Clear All
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 54
The pH of ammonium phosphate solution, if $$pK_a$$ of phosphoric acid and $$pK_b$$ of ammonium hydroxide are 5.23 and 4.75 respectively, is ______
789
456
123
0.-
Clear All
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 55
In mildly alkaline medium, thiosulphate ion is oxidized by $$MnO_4^-$$ to 'A'. The oxidation state of sulphur in 'A' is ______
789
456
123
0.-
Clear All
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 56
The number of octahedral voids per lattice site in a lattice is ______. (Rounded off to the nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 57
When 12.2 g of benzoic acid is dissolved in 100 g of water, the freezing point of solution was found to be $$-0.93$$ °C ($$K_f(H_2O) = 1.86$$ K kg mol$$^{-1}$$). The number (n) of benzoic acid molecules associated (assuming 100% association) is ______.
789
456
123
0.-
Clear All
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 58
Emf of the following cell at 298 K in V is $$x \times 10^{-2}$$
$$Zn|Zn^{2+}(0.1M)||Ag^+(0.01M)|Ag$$
The value of $$x$$ is ______ (Rounded off to the nearest integer)
[Given: $$E^\theta_{Zn^{2+}/Zn} = -0.76$$ V; $$E^\theta_{Ag^+/Ag} = +0.80$$ V; $$\frac{2.303RT}{F} = 0.059$$]
789
456
123
0.-
Clear All
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 59
If the activation energy of a reaction is 80.9 kJ mol$$^{-1}$$, the fraction of molecules at 700 K, having enough energy to react to form products is $$e^{-x}$$. The value of $$x$$ is (Rounded off to the nearest integer) [Use R = 8.31 J K$$^{-1}$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 26th February 2021 Shift 2 - Chemistry - Question 60
The number of stereo isomers possible for $$[Co(ox)_2(Br)(NH_3)]^{2-}$$ is ______.
[ox = oxalate]
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




